Acetophenone: Applications, Pharmacokinetics and Synthesis
Introduction
Acetophenone[1] is an organic compound with the chemical formula C8H8O. It is a colorless liquid with a sweet, flowery odor that is used as a solvent and a precursor in the synthesis of many other organic compounds. Acetophenone is also used as a flavoring agent in foods and as a fragrance in perfumes and soaps. In addition, it is a key component in the production of various pharmaceuticals, including anticonvulsants, hypnotics, and analgesics.

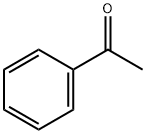
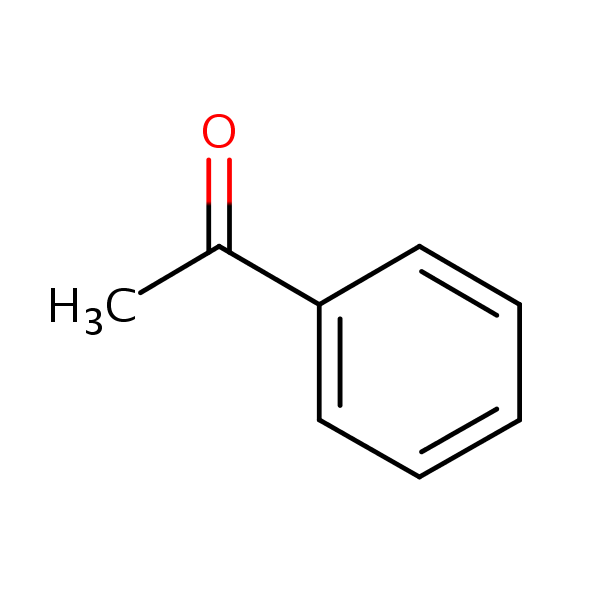
Figure 1 chemical formula of Acetophenone
Applications
There are some specific examples of the application of Acetophenone in different industries[2]:
1. As a fragrance ingredient: Acetophenone is used as a fragrance ingredient in various products such as soaps, detergents, lotions, and perfumes. It has a sweet, floral, and fruity fragrance that can be blended with other fragrances to create complex scents.
2. In the production of polycarbonate resins: Acetophenone is used as a raw material in the production of polycarbonate resins. These resins have excellent optical clarity, impact resistance, and heat resistance, making them ideal for use in eyeglass lenses, automotive parts, and electronic components.
3. In the manufacture of pharmaceuticals: Acetophenone is used to produce various pharmaceuticals such as antihistamines, anti-inflammatory drugs, and muscle relaxants. For example, it is used in the synthesis of phenytoin, which is an anticonvulsant drug[3].
4. As a solvent in organic synthesis: Acetophenone is a versatile solvent that is used in the synthesis of many organic compounds. It is particularly useful in reactions involving Grignard reagents, which are commonly used in organic synthesis.
5. As a flavoring agent in food: Acetophenone is used as a flavoring agent in baked goods, candies, and chewing gum. It has a sweet and slightly nutty flavor that is similar to almond extract.
6. In the production of epoxy resins: Acetophenone is used as a curing agent for epoxy resins. Epoxy resins are widely used in adhesives, coatings, and composites due to their excellent mechanical properties and chemical resistance[4].
Overall, acetophenone has many specific applications in various industries due to its unique chemical properties and versatility.
Pharmacokinetics
The pharmacokinetics of acetophenone refers to how the body processes this substance after administration or exposure.
Absorption: Acetophenone can be absorbed through inhalation, ingestion, and skin contact. After inhalation, it is rapidly absorbed into the bloodstream through the lungs. When ingested, its absorption occurs in the gastrointestinal tract. It has been reported that absorption through the skin is limited.
Distribution: After absorption, acetophenone is distributed throughout the body primarily through the bloodstream. Its distribution is quite rapid because it is a small molecule with low molecular weight. However, the extent of distribution into tissues is unknown.
Metabolism: Acetophenone undergoes metabolism mainly in the liver. The primary metabolic pathway is oxidation, which converts acetophenone to phenylacetate. Phenylacetate can then be converted to hippurate by conjugation with glycine. Both phenylacetate and hippurate are excreted in urine.
Elimination: Acetophenone and its metabolites are eliminated mainly via urine. The elimination half-life of acetophenone in humans is not well established, but some studies suggest that it may be relatively short.
Factors affecting pharmacokinetics: Factors such as age, sex, genetics, and concurrent use of other substances may affect the pharmacokinetics of acetophenone. For example, individuals who are slow metabolizers of acetophenone due to genetic factors may be more susceptible to toxicity from chronic exposure[5].
Synthesis
Acetophenone is an important organic compound that can be synthesized by several methods. One of the most common methods is the Friedel-Crafts acylation reaction, which involves the reaction of benzene with acetyl chloride in the presence of a Lewis acid catalyst such as aluminum chloride. The resulting intermediate undergoes rearrangement to form acetophenone[6-11].
Another method for synthesizing acetophenone is the oxidation of ethylbenzene using air or oxygen in the presence of a copper or manganese catalyst. This process yields acetophenone as one of the major products along with acetone.
Acetophenone can also be prepared via the Perkin reaction, which involves the condensation of benzaldehyde with acetic anhydride in the presence of an acidic catalyst such as sulfuric acid.
Overall, acetophenone is a versatile compound that can be synthesized by several methods, each offering specific advantages in terms of yield, purity, and ease of synthesis.
References
[1] Tong Qingjun, Fan Lei, Li Pingping. Synthesis of Ag2C2O4 Catalyst and Its Application of Ethylbenzene Oxidation to Acetophenone [J]. Industrial Catalysis, 2022,30(08):66-71.
[2] Geohagen Brian,Zeldin Elizabeth,Reidy Kimberly,Wang Tao,Gavathiotis Evripidis,Fishman Yonatan I.,LoPachin Richard,Loeb David M.,Weiser Daniel A.. Acetophenone protection against cisplatin-induced end-organ damage[J]. Translational Oncology,2023,27.
[3]Shen Yanhong,Li Baoli,Xu Hu,Zhang Guoqiang. Design, synthesis, and biological evaluation of acetophenone derivatives as dual binding acetylcholinesterase inhibitors.[J]. Die Pharmazie,2013,68(5).
[4]Lagaditis Paraskevi O,Lough Alan J,Morris Robert H. Iron complexes for the catalytic transfer hydrogenation of acetophenone: steric and electronic effects imposed by alkyl substituents at phosphorus.[J]. Inorganic chemistry,2010,49(21).
[5]Du Kaicheng,Zhang Zhiqi,Jing Di,Wang Yumeng,Li Xingyao,Meng Dali. Diterpene glycosides, acetophenone glycosides and tannins from polar extracts of the root of Euphorbia fischeriana with cytotoxicity and antibacterial activities.[J]. Phytochemistry,2022,203.
[6] Wang Yifan, Zhou Weiyou, Fang Xiao, Chen Qun. Catalytic Oxidation of Ethylbenzene to Synthesis of Acetophenone by Magnesium Aluminum Hydrotalcite Supported by MnO_4~-[J]. Applied Chemistry, 2012,29(09):1017-1022.
[7] Li Guixian, Gao Yunyan, Zhang Xuemei, Song Weiwei, Wang Guang, Wang Tianfeng. Synthesis of acetophenone by one-step oxidation of ethylbenzene with tert-butyl hydroperoxide[J]. Petrochemical Industry, 2011,40(06):599-602.
[8] Song Hua, Wang Yuanyuan, Liu Siyu, Zhang Jinzhao. Green oxidation synthesis of acetophenone [J]. Chemical Bulletin, 2009, 72(06): 544-548. DOI: 10.14159/j.cnki.0441-3776.2009. 06.008.
[9] Song Hua, Wang Yuanyuan, Liu Siyu, Zhang Jinzhao, Sun Xinglong. Manganese sulfate catalyzed oxygen oxidation of ethylbenzene to acetophenone[J]. Chemical Industry Progress, 2009,28(06):971-974.
[10] Song Hua, Wang Yuanyuan, Liu Siyu, Zhang Jinzhao. Research progress in the green production process of synthetic acetophenone [J]. Chemical Production and Technology, 2008 (05): 42-45+8.
[11]Dawei Chen,Ridao Chen,Kebo Xie,Yangyang Duan,Jungui Dai. Production of acetophenone C-glucosides using an engineered C-glycosyltransferase in Escherichia coli[J]. Tetrahedron Letters,2018,59(19).
Related articles And Qustion
See also
Lastest Price from Acetophenone manufacturers

US $100.00/KG2025-04-21
- CAS:
- 98-86-2
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 200TON

US $99.00-66.00/kg2025-04-21
- CAS:
- 98-86-2
- Min. Order:
- 0.0010000000474974513kg
- Purity:
- 99%
- Supply Ability:
- 5000