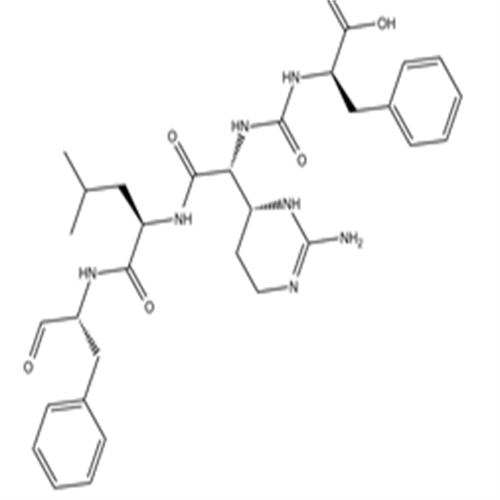
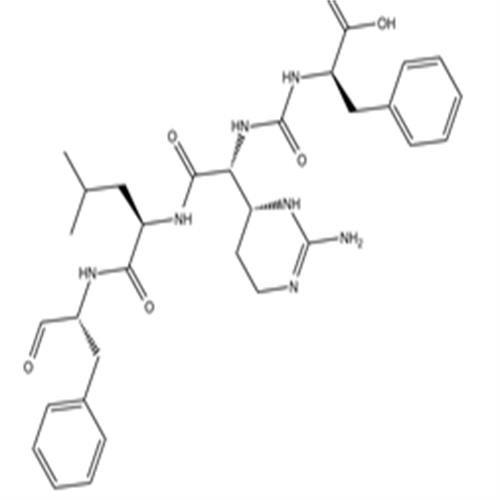
9076-44-2Chymostatin
Chymostatin
9076-44-2
9076-44-2
询价
1盒
起订
上海 更新日期:2025-11-06
产品详情:
- 中文名称:
- Chymostatin
- 英文名称:
- Chymostatin
- CAS号:
- 9076-44-2
- 品牌:
- 一研
- 保存条件:
- Store at -20°C
- 产品类别:
- 抑制剂
- Cas:
- 9076-44-2
公司简介
上海一研生物科技有限公司Shanghai yiyan bio-technology Co. Ltd.主要从事免疫学、分子生物学和常规生化试剂等为一体的科研产品销售企业,公司自成立以来,秉承""全心全意服务于科研工作者""的企业理念,立足生物科技领域,运用生物技术和科研试剂,发展现代生物科技,为各类大中小医院及其它医疗机构、高等院校、科研院所、企事业单位提供优质的产品,服务生物科技领域的科学研究人员。
公司具有对普通货物、冷藏及冷冻仓库的存储、包装及运输能力。
公司将始终坚持信誉立业、以人为本、质量保证、诚信服务的宗旨,不断拼搏,开拓进取,与各界朋友携手共创美好未来。
| 成立日期 | (12年) |
| 注册资本 | 100 |
| 员工人数 | 50-100人 |
| 年营业额 | ¥ 100万以内 |
| 经营模式 | 工厂,试剂 |
| 主营行业 | 生化试剂,抗体,细胞培养,分子生物学,免疫安全 |
Chymostatin相关厂家报价
-
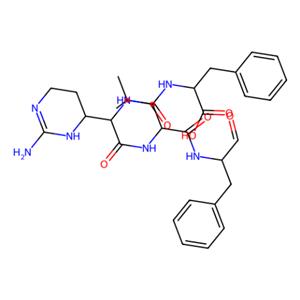
- aladdin 阿拉丁 C113165 胰凝乳蛋白酶抑制剂 9076-44-2 ≥95%
- 上海阿拉丁生化科技股份有限公司 VIP
- 2026-05-14
- ¥432.90
-
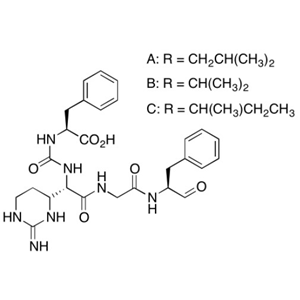
- CATO_胰凝乳蛋白酶抑制剂_9076-44-2_97%
- 广州佳途科技股份有限公司 VIP
- 2026-04-22
- 询价
-
- 胰凝乳蛋白酶抑制剂 9076-44-2
- 宝鸡缔都医药化工有限公司 VIP
- 2026-03-24
- 询价
-

- 促凝素
- 湖北艾普蒂生物工程有限公司
- 2023-08-09
- ¥1200