| CAS: | 15956-28-2 |
| MF: | C8H12O8Rh2 |
| MW: | 441.99 |
| EINECS: | 240-084-8 |
| Product Categories: | Catalysts for Organic Synthesis;Classes of Metal Compounds;Homogeneous Catalysts;Metal Complexes;Rh (Rhodium) Compounds;Rh;Synthetic Organic Chemistry;Transition Metal Compounds;metal acetate salt;chemical reaction,pharm,electronic,materials |
| Mol File: | 15956-28-2.mol |
 |
|
| Rhodium(II) acetate dimer Chemical Properties |
| Melting point | 205 °C |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | Methanol + Water (Slightly) |
| form | powder |
| color | Green to dark green |
| Water Solubility | Soluble in aqueous solution like water. Slightly soluble in ethanol |
| Hydrolytic Sensitivity | 0: forms stable aqueous solutions |
| Exposure limits | ACGIH: TWA 0.01 mg/m3
NIOSH: IDLH 2 mg/m3; TWA 0.001 mg/m3 |
| Stability: | Hygroscopic |
| InChIKey | ANSVJOOIUURYRN-UHFFFAOYSA-N |
| Rhodium(II) acetate dimer Usage And Synthesis |
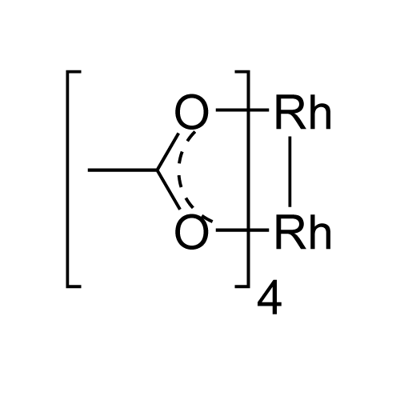
| Chemical Properties | Rhodium(II) acetate dimer is Emerald Green Powder. Soluble in methanol. It is soluble in water, while the relative solubility of copper acetate in water is small.
 |
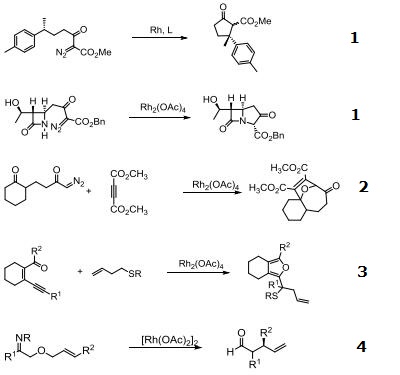
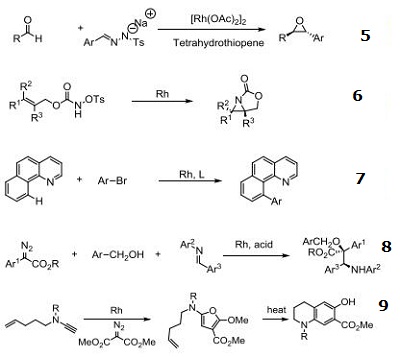
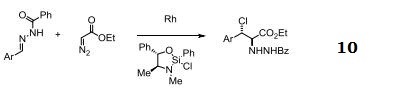
| Uses | Rhodium (II) Acetate Dimer is a effective catalyst for ylide formation and it is also catalyst for cyclopropanation of alkenes, oxidation of alcohols, cyclization reactions involving -diazo carbonyl groups, insertion into C-H and X-H bonds (X-H is NH or SH or OH). More reactive and useful in differentiating ribonucleosides and deoxynucleosides. It is also used in functionalizing fullerenes into polymers. Efficient catalyst for hydrogen transfer from 2-propanol to cyclohexanone and other unsaturated compounds. |
| Application | Rhodium (II) Acetate Dimer is used in the preparation of molybdenum triisopropylbenzoate isonicotinate which maintains ambivalent properties. |
| Preparation | Rhodium(II) acetate dimer synthesis: A suspension of 10.0g. of Rh(OH)3.H20 in 400 ml. of glacial acetic acid dissolved upon refluxing for 18 hr. to give a deep emerald-green solution. Most of the acetic acid was evaporated on a steam bath; remaining traces mere removed by heating the residue at 120° for 1hr. The residue was then extracted with boiling acetone until the extract was colorless rather than bluish green. The extract was quickly passed through a fritted glass filter, concentrated on a steam bath to 1/3 its original volume, and placed in the freezing compartment of a refrigerator for 18hr. The resulting large dark green crystals were collected on a filter, washed with small portions of ice-cold acetone, and dried at 110°. When first collected, the product is the weak adduct [Rh(OOCCH3)2(CH3)2C0]2; however, acetone is lost fairly readily at room temperature and immediately in the drying oven; yield, 6.2g. (48%). |
| Reactions | Catalyst for insertion into C-H and X-H bonds.
Catalyst for Ylide generation.
Doyle Kirmse Reaction of Allylic Sulfides with Diazoalkane.
Claisen rearrangement.
Epoxides from aldehydes.
Synthesis of aziridines from allylic N-tosyloxycarbamates.
Rh/NHC catalyzed direct intermolecular arylation of C-H bonds.
Chiral Bronsted acid-Rh catalyzed three component reactions of diazo compounds with alcohols and imines.
Rh-catalyzed cyclopropenations of ynamides.
Tandem asymmetric aza-Darzens/ring-opening reactions.
 |
| Purification Methods | Dissolve 5g of the salt in boiling MeOH (ca 600mL) and filter. Concentrate it to 400mL and chill overnight at ca 0o to give dark green crystals of the MeOH adduct. Concentration of the mother liquors gives a further crop of [Rh(OAc)2]2.2MeOH. The adduct is then heated at 45o in a vacuum for 2hours (all MeOH is lost) to leave the emerald green crystals of the actetate. [Legzdins et al. J Chem Soc (A) 3322 1970.] Alternatively dissolve the acetate in glacial AcOH and reflux for a few hours to give an emerald green solution. Evaporate most of the AcOH on a steam bath, then heat the residue at 120o/1hour. Extract the residue with boiling Me2CO. Filter, concentrate to half its volume and keep at 0o/18hours. Collect the crystals, wash them with ice cold Me2CO and dry them at 110o. It is soluble in most organic solvents with which it forms adducts including Me3N and Me2S and gives solutions with different colours varying from green to orange and red. [UV: Johnson et al. Inorg Chem 2 960 1963, Beilstein 1 H 124.] |
Packing &shipping&Payment
Packing:25kg/drum
Shipping:by sea or by air
Payment:T/T,western union,moneygram
Packaging Details drum
Port:Tianjin
Lead Time :
| Quantity(Kilograms) | 1 - 10000 | >10000 |
| Est. Time(days) | 5 | To be negotiated |

 Company information
Company information
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in 4'-Methylacetophenone,Levamisole hydrochloride ,N-Methylformamide and other chemical reagents research and development production enterprises. Our business covers more than 30 countries, most of the big customers come from Europe, America and other countries in the world, we can guarantee the quality and price. In recent decades, with the efforts of all employees, we have established many cooperative companies in shandong, henan, guangdong and other places. Our corporate purpose is based on the market, enhance the strength, take the road of scientific and environmental sustainable development, relying on the country. Technology r & d center, increase the investment in r & d, based on the domestic market, expand the international market, manufacturing quality products, sincere service to the society, into a modern, ecological, scientific and technological enterprise world.
 Advantage
Advantage
In stock

 Japan
Japan











 Company information
Company information Advantage
Advantage


