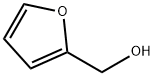
| Description |
Furfuryl alcohol is clear colorless organic liquid having a furan substituted with a hydroxymethyl group. It is primarily used for the synthesis of furans resins which are used in thermoset polymer matrix composites, cements, adhesive and coatings. It plays an essential role in the production of foundry sand binder and has long been used to produce cores and molds for metal casting. Other applications include as a fuel and wood treatment. In industry, it is manufactured through either direct reduction of furfural, or through the disproportionation via the Cannizaro reaction in NaOH solution. The basic raw materials for its manufacturing are waste vegetable materials such as rice hulls, sugar cane bagasse, oat hulls or corncobs. |
| References |
https://en.wikipedia.org/wiki/Furfuryl_alcohol
http://www.furan.com/furfuryl_alcohol_applications.html |
| Chemical Properties |
clear yellow liquid |
| Uses |
Furfuryl Alcohol has been obtained by yeast reduction of furfural. Furfuryl Alcohol is used as solvent and in the manufacturing of wetting agents, resins. |
| Definition |
ChEBI: A member of the class of furans bearing a hydroxymethyl substituent at the 2-position. |
| Uses |
Solvent; manufacture of wetting agents, resins. |
| General Description |
A clear colorless liquid. Flash point 167°F. Boiling point 171°F. Denser than water. Contact may irritate skin, eyes and mucous membranes. May be toxic by ingestion and skin contact and moderately toxic by inhalation. |
| Air & Water Reactions |
Slightly soluble in water. |
| Hazard |
May react explosively with mineral acids and some organic acids. Toxic by inhalation and skin absorption. Approved for food products. Toxic by skin absorption. |
| Health Hazard |
Inhalation causes headache, nausea, and irritation of nose and throat. Vapor irritates eyes; liquid causes inflammation and corneal opacity. Contact of skin with liquid causes dryness and irritation. Ingestion causes headache, nausea, and irritation of mouth and stomach. |
| Fire Hazard |
Combustible material: may burn but does not ignite readily. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. Runoff may pollute waterways. Substance may be transported in a molten form. |
| Safety Profile |
Poison by ingestion, skin contact, and subcutaneous routes.Moderately toxic by inhalation and intraperitoneal routes. Mutation data reported. An eye irritant. Flammable when exposed to heat or flame; can react with oxidtzing materials. Moderate explosion hazard when exposed to heat or flame. Reacts violently with acids (e.g., formic acid, cyanoacetic acid + heat). Ignites on contact with 85% hydrogen peroxide. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and fumes. |
| Purification Methods |
Distil it under reduced pressure to remove tarry material, shake with aqueous NaHCO3, dry it with Na2SO4 and fractionally distil it under reduced pressure from Na2CO3. It can be further dried by shaking with Linde 5A molecular sieves. [Beilstein 17/3 V 338.] |

 China
China