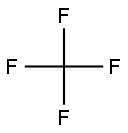
| Chemical Properties |
colourless gas |
| Uses |
Low tempereture refrigerant; gaseous insulator. |
| General Description |
Tetrafluoromethane is a colorless nonflammable gas. Carbon tetrafluoride is shipped as a liquid under pressure. Carbon tetrafluoride may be narcotic at high concentrations. Under prolonged exposure to fire or heat the containers may rupture violently and rocket. Carbon tetrafluoride is used as a refrigerant. |
| Reactivity Profile |
The reaction of aluminum with various halogenated hydrocarbons produces a self-sustaining reaction with sufficient heat to melt aluminum pieces, examples of other halogenated hydrocarbons are fluorotrichloromethane, dichlorodifluoromethane, chlorodifluoromethane, tetrafluoromethane. The vigor of the reaction appears to be dependent on the combined degree of fluorination and the vapor pressure [Chem. Eng. News 39(27):44. 1961]. |
| Hazard |
Toxic by inhalation. |
| Health Hazard |
Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating, corrosive and/or toxic gases. |
| Fire Hazard |
Some may burn but none ignite readily. Containers may explode when heated. Ruptured cylinders may rocket. |
| Purification Methods |
Purify CF4 by repeated passage over activated charcoal at solid-CO2 temperatures. Traces of air are removed by evacuating while alternately freezing and melting. Alternatively, liquefy CF4 by cooling in liquid air and then fractionally distil it under vacuum. (The chief impurity originally present is probably CF3Cl). Use brass equipment. It is non-flammable. [Beilstein 1 H 59, 1 I 8, 1 II 11, 1 III 35, 1 IV 26.] |

 China
China