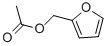| Description |
Furfuryl acetate is the ester formed by the esterification between furfuryl alcohol and acetate. It can be used as a food spices and flavoring ingredient. It can also be used as the intermediate of dye, resin and spices. It is found in alcoholic beverage. Furfuryl acetate is present in wheat bread, crisp bread, roasted onion, pork liver, beer, rum, cocoa and coffee. |
| References |
Kim, You Sun, S. S. Lee, and M. W. Oh. "Halogen Containing Heterocyclic Compounds (Part III) Chlorination of Furfuryl Acetate in Presence of Acid and Lewis Acids." Soil Biology & Biochemistry 924.1(1970):263-270.
Harayama, Koichi, F. Hayase, and H. Kato. "Contribution to Stale Flavor of 2-Furfuryl Ethyl Ether and Its Formation Mechanism in Beer." Bioscience Biotechnology & Biochemistry 59.6(1995):1144-1146.
Antón, Víctor, et al. "Thermophysical Characterization of Furfuryl Esters: Experimental and Modeling." Energy & Fuels (2017). |
| Chemical Properties |
Colorless to light yellow liqui |
| General Description |
Colorless to clear yellow or orange liquid with a pungent odor. |
| Air & Water Reactions |
Slightly water soluble. |
| Reactivity Profile |
Furfuryl acetate is an ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides. Furfuryl acetate reacts with strong oxidizing agents, strong acids, strong bases and strong reducing agents. Furfuryl acetate reacts violently with cyanoacetic acid, formic acid, mineral acids, nitric acid and (nitric acid + N204 + sulfuric acid). |
| Fire Hazard |
Furfuryl acetate is combustible. |

 China
China




 Product Information:
Product Information: