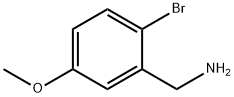
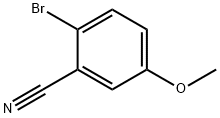
Step A: Synthesis of 1-(2-bromo-5-methoxyphenyl)methanamine
2-Bromo-5-methoxybenzonitrile (10.0 g, 47.2 mmol) was dissolved in dry tetrahydrofuran (100 mL) in a flame-dried flask and cooled in an ice bath. A solution of borane-tetrahydrofuran complex (75 mL, 75 mmol, 1.0 M) was added slowly and dropwise over 30 min. The reaction mixture was stirred overnight at room temperature and then the reaction was slowly quenched with ice water and saturated aqueous sodium carbonate solution. Tetrahydrofuran was removed by distillation under reduced pressure and the residue was extracted with ethyl acetate (EtOAc) several times (3 times). The organic layers were combined, washed with water, dried over anhydrous sodium sulfate and concentrated under reduced pressure to give the crude product. The crude product was purified by silica gel column chromatography to afford the target product 1-(2-bromo-5-methoxyphenyl)methanamine as a white powder (3.65 g, 36% yield). m/z = 216.9, 219.0 for C8H10BrNO ([M+H]+) by LCMS analysis.