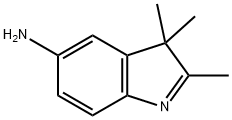
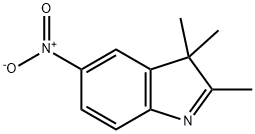
General procedure for the synthesis of 5-amino-2,3,3-trimethyl-3H-indole (product B) from 5-nitro-2,3,3-trimethylindole (product A):
Example of implementation:
A mixture of 6.6 g of Product A, 43.7 g of stannous chloride dihydrate and 215 mL of hydrochloric acid was heated to reflux for 2 hours. Upon completion of the reaction, it was cooled to room temperature and filtered. The collected solid was dissolved in 130 mL of water and neutralized to pH neutral with 20% sodium carbonate solution. The precipitate precipitated was filtered, washed several times with water and subsequently dried under vacuum in the presence of phosphoric acid. 5.4 g of product B was finally obtained in 96% yield.
[1] Patent: US2012/45851, 2012, A1. Location in patent: Page/Page column 14; 18
[2] Organic and Biomolecular Chemistry, 2012, vol. 10, # 4, p. 710 - 715
[3] Chemistry of Heterocyclic Compounds (New York, NY, United States), 1984, vol. 20, # 9, p. 976 - 977
[4] Khimiya Geterotsiklicheskikh Soedinenii, 1984, vol. 20, # 9, p. 1198 - 1199
[5] Journal of Physical Chemistry, 1985, vol. 89, # 18, p. 3941 - 3946