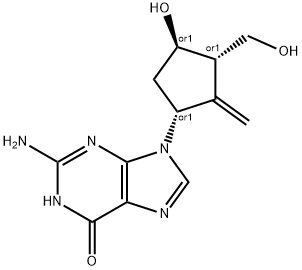
ChEBI: Entecavir (anhydrous) is guanine substituted at the 9 position by a 4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl group. A synthetic analogue of 2'-deoxyguanosine, it is a nucleoside reverse transcriptase inhibitor with selective antiviral activity against hepatitis B virus. Entecavir is phosphorylated intracellularly to the active triphosphate form, which competes with deoxyguanosine triphosphate, the natural substrate of hepatitis B virus reverse transcriptase, inhibiting every stage of the enzyme's activity, although it has no activity against HIV. It is used for the treatment of chronic hepatitis B. It has a role as an EC 2.7.7.49 (RNA-directed DNA polymerase) inhibitor and an antiviral drug. It is a member of 2-aminopurines, an oxopurine, a primary alcohol and a secondary alcohol.