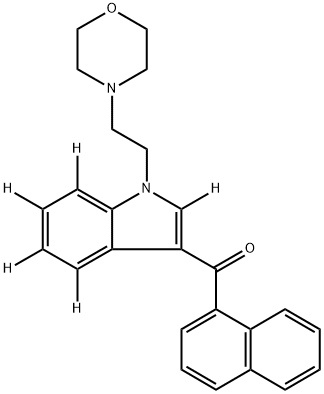
JWH 200-
d
5 contains five deuterium atoms at the 2', 4' ,5', 6', and 7' positions. It is intended for use as an internal standard for the quantification of JWH 200 by GC-
or LC-
mass spectrometry. JWH 200 is an aminoalkylindole that acts as a cannabinoid (CB) receptor ligand. It binds to the CB
1 receptor with high-
affinity (IC
50 = 7.8-
42 nM).
1,2 The effects of JWH 200 in locomotor activity, tail-
flick latency, hypothermia, and ring-
immobility tests are comparable or superior to Δ
9-
THC or WIN 55,212.
3 It potently inhibits the contraction of electrically-
stimulated mouse vas deferens (IC
50 = 3.7-
6.0 nM).
4,5
1. Eissenstat, M.A., Bell, M.R., D'Ambra, T.E., et al. Aminoalkylindoles: Structure-activity relationships of novel cannabinoid mimetics J. Med. Chem. 38,3094-3105(1995).
2. Huffman, J.W., Mabon, R., Wu, M.J., et al. 3-indolyl-1-naphthylmethanes: New cannabimimetic indoles provide evidence for aromatic stacking interactions with the CB1 cannabinoid receptor Bioorgan. Med. Chem. 11(4),539-549(2003).
3. Compton, D.R., Gold, L.H., Ward, S.J., et al. Aminoalkylindole analogs: Cannabimimetic activity of a class of compounds structurally distinct from D 9-tetrahydrocannabinol J. Pharmacol. Exp. Ther. 263(3),1118-1126(1992).
4. Pacheco, M., Childers, S.R., Arnold, R., et al. Aminoalkylindoles: Actions on specific G-protein-linked receptors J. Pharmacol. Exp. Ther. 257(1),170-183(1991).
5. Bell, M.R., D'Ambra, T.E., Kumar, V., et al. Antinociceptive (aminoalkyl) indoles J. Med. Chem. 34,1099-1110(1991).