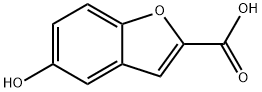
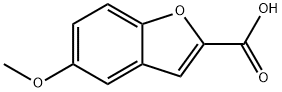
Step 1: Preparation of 5-hydroxybenzofuran-2-carboxylic acid. 5-Methoxybenzofuran-2-carboxylic acid (1.0 g, 5.2 mmol) was dissolved in anhydrous dichloromethane (25 ml) and the solution was cooled to -78°C. A 1 M solution of boron tribromide dichloromethane (15.6 ml) was added slowly and dropwise under nitrogen protection. The reaction mixture was gradually warmed to room temperature and stirred continuously at this temperature for 4 hours. Upon completion of the reaction, the reaction was quenched with aqueous ammonium chloride solution (20 ml) and subsequently extracted with ethyl acetate. The organic layer was washed with water and dried over anhydrous sodium sulfate. After treatment, 0.9 g (100% yield) of the crude product of 5-hydroxybenzofuran-2-carboxylic acid was obtained.1H NMR (CD3OD) δ = 6.84 (dd, 1H), 6.95 (d, 1H), 7.17 (s, 1H), 7.32 (d, 1H).