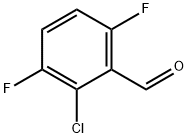
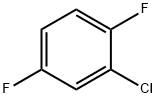
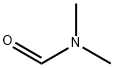
Example 152A Synthesis of 2-chloro-3,6-difluorobenzaldehyde: 2-chloro-1,4-difluorobenzene (1.78 g, 12.0 mmol) was dissolved in tetrahydrofuran (50 mL) and the solution was cooled to -78°C. Lithium diisopropylammonium (1.8 M in tetrahydrofuran, 7.3 mL, 13.2 mmol) was added slowly with stirring. The reaction temperature was maintained at -78 °C and stirring was continued for 30 min. Subsequently, N,N-dimethylformamide (1.05 g, 14.4 mmol) was added and stirring was continued at the same temperature for 15 min. After the reaction was completed, acetic acid (3 mL) and water (100 mL) were added and the mixture was gradually warmed up to room temperature. Extraction was carried out with ethyl acetate and the organic phases were combined and washed sequentially with 1 M hydrochloric acid solution and saturated saline. The organic layer was dried with anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to afford the target product 2-chloro-3,6-difluorobenzaldehyde (1.51 g, 71% yield). The product was analyzed by GC-MS (Method 1G): retention time Rt = 3.25 min, mass spectrum (ESI positive ion mode): m/z = 177 [M + H]+.