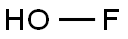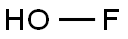Hypofluoric acid is a relatively novel oxidant. The OF bond is unstable; oxygen is connected to fluorine, which is more electronegative than oxygen, so that the oxygen in it is biased to be positively charged. It is a highly electrophilic oxygen-donating reagent (the oxygen in hypofluoric acid) and strong applicability.