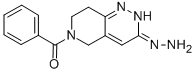
Usage And Synthesis
(a) 6-Carbethoxy-5,6,7,8-tetrahydro-3(2H)pyrido[4,3-c]pyridazinone:
Produced from 450.5 g of 2,3,4,4a,5,6,7,8-octahydro-3-oxo-6-
pyrido[4,3c]pyridazinecarboxylic acid ethyl ester and 320 g of bromine. The
bromine is added dropwise to a boiling solution of the ester in 200 cc of
chloroform over one hour and the mixture is stirred for another hour at the
same temperature. 1 kg of ice water is added to the mixture, the chloroform
portion is separated, and the acid aqueous phase is again extracted with 500
cc of chloroform. The semicrystalline crude product obtained after
concentrating the chloroform phase, is recrystallized with 250 cc of absolute
ethanol, melting point 165°C to 168°C (decomp.).
A solution of 223.2 g of 6-carbethoxy-5,6,7,8-tetrahydro-3(2H)pyrido[4,3- c]pyridazinone in 1 liter of concentrated hydrochloric acid is heated to the boil at reflux for 22 hours while stirring. The mixture is concentrated in a vacuum, and the resulting crude crystalline hydrochloride of 5,6,7,8-tetrahydro- 3(2H)pyrido[4,3-c]pyridazinone, having a melting point of 307°C to 310°C (decomposed from methanol), is suspended in 0.75 liter of methanol, and 0.4 liter of triethylamine is slowly added to the suspension. After stirring for 15 minutes and cooling the violet suspension, the crude base is obtained. 25 g of the crude base are recrystallized from 300 cc of methanol, mixed with 10 cc of concentrated ammonia and 40 cc of water, with the addition of a small amount of coal. 5,6,7,8-Tetrahydro-3(2H)pyrido[4,3-c]pyridazinone has a melting point of 223°C to 225°C (decomp.).
(b) 3-Chloro-5,6,7,8-tethydropyrido[4,3-c]pyridazine: Produced from 30.3 g of 5,6,7,8-tetrahydro3(2H)pyrido[4,3-c]pyridazinone suspended in 250 cc of phosphorus oxychloride. The suspension is heated to the boil while stirring. The resulting solution is stirred for 1 hour at the boil and then concentrated to an oil in a vacuum. 150 cc of ice water and 40 cc of concentrated ammonia solution are added to this oil, and the mixture is extracted twice with a total of 300 cc of chloroform. The chloroform phase is concentrated in a vacuum.
(c) The crude unstable base is converted into the maleate for working up. This is effected by boiling 24.8 g of the base in 150 cc of methanol with 17.5 g ofmaleic acid. Upon cooling the solution, the crude maleate is obtained, which is recrystallized from methanol with the addition of a small amount of coal. 3- Chloro-5,6,7,8-tetrahydropyrido[4,3-c]pyridazine maleate has a melting point of 162°C to 164°C (decomp.).
A mixture of 12.6 g of benzoyl chloride in 100 cc of ethylene chloride is added dropwise to a suspension of 25.6 g of 3-chloro-5,6,7,8-tetrahydropyrido[4,3- c]pyridazine maleate in 250 cc of ethylene chloride and 21.8 g of triethylamine within 18 minutes at room temperature while stirring. The mixture is stirred at room temperature for a further 14 hours, 200 cc of water are added, the organic phase is separated and concentrated to an oil in a vacuum. Upon adding ether/dimethoxyethane to this oil, crude 6-benzoyl-3- chloro-5,6,7,8-tetrahydropyrido[4,3-c]pyridazine is obtained. After recrystallization from absolute ethanol with the addition of a small amount of coal, the compound has a melting point of 125°C to 127°C (decomp.). Displacement of the halogen with hydrazine leads to the formation of endralazine.
A solution of 223.2 g of 6-carbethoxy-5,6,7,8-tetrahydro-3(2H)pyrido[4,3- c]pyridazinone in 1 liter of concentrated hydrochloric acid is heated to the boil at reflux for 22 hours while stirring. The mixture is concentrated in a vacuum, and the resulting crude crystalline hydrochloride of 5,6,7,8-tetrahydro- 3(2H)pyrido[4,3-c]pyridazinone, having a melting point of 307°C to 310°C (decomposed from methanol), is suspended in 0.75 liter of methanol, and 0.4 liter of triethylamine is slowly added to the suspension. After stirring for 15 minutes and cooling the violet suspension, the crude base is obtained. 25 g of the crude base are recrystallized from 300 cc of methanol, mixed with 10 cc of concentrated ammonia and 40 cc of water, with the addition of a small amount of coal. 5,6,7,8-Tetrahydro-3(2H)pyrido[4,3-c]pyridazinone has a melting point of 223°C to 225°C (decomp.).
(b) 3-Chloro-5,6,7,8-tethydropyrido[4,3-c]pyridazine: Produced from 30.3 g of 5,6,7,8-tetrahydro3(2H)pyrido[4,3-c]pyridazinone suspended in 250 cc of phosphorus oxychloride. The suspension is heated to the boil while stirring. The resulting solution is stirred for 1 hour at the boil and then concentrated to an oil in a vacuum. 150 cc of ice water and 40 cc of concentrated ammonia solution are added to this oil, and the mixture is extracted twice with a total of 300 cc of chloroform. The chloroform phase is concentrated in a vacuum.
(c) The crude unstable base is converted into the maleate for working up. This is effected by boiling 24.8 g of the base in 150 cc of methanol with 17.5 g ofmaleic acid. Upon cooling the solution, the crude maleate is obtained, which is recrystallized from methanol with the addition of a small amount of coal. 3- Chloro-5,6,7,8-tetrahydropyrido[4,3-c]pyridazine maleate has a melting point of 162°C to 164°C (decomp.).
A mixture of 12.6 g of benzoyl chloride in 100 cc of ethylene chloride is added dropwise to a suspension of 25.6 g of 3-chloro-5,6,7,8-tetrahydropyrido[4,3- c]pyridazine maleate in 250 cc of ethylene chloride and 21.8 g of triethylamine within 18 minutes at room temperature while stirring. The mixture is stirred at room temperature for a further 14 hours, 200 cc of water are added, the organic phase is separated and concentrated to an oil in a vacuum. Upon adding ether/dimethoxyethane to this oil, crude 6-benzoyl-3- chloro-5,6,7,8-tetrahydropyrido[4,3-c]pyridazine is obtained. After recrystallization from absolute ethanol with the addition of a small amount of coal, the compound has a melting point of 125°C to 127°C (decomp.). Displacement of the halogen with hydrazine leads to the formation of endralazine.
Preparation Products And Raw materials
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine