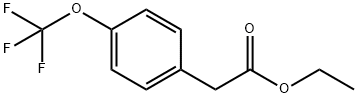
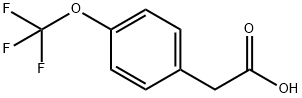
a) Concentrated sulfuric acid (0.484 mL, 9.08 mmol) was added slowly and dropwise to a solution of 2-(4-(trifluoromethoxy)phenyl)acetic acid (2.0 g, 9.08 mmol) in anhydrous ethanol (20 mL), which was subsequently heated to reflux and held for 5 hours. Upon completion of the reaction, the reaction mixture was concentrated under reduced pressure to remove the solvent and then diluted with ethyl acetate (100 mL). The diluted solution was washed sequentially with saturated sodium bicarbonate solution and saturated brine to remove acidic impurities and inorganic salts. The organic layer was dried over anhydrous magnesium sulfate, filtered and concentrated again under reduced pressure to give the crude product. The crude product was purified by column chromatography (eluent: ethyl acetate/hexane, gradient elution, 0/100 to 20/80) to finally obtain ethyl 2-(4-(trifluoromethoxy)phenyl)acetate (2.06 g, 91% yield) as a yellow oil.