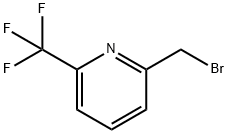
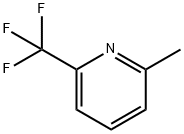
General procedure for the synthesis of 2-(bromomethyl)-6-(trifluoromethyl)pyridine using 2-methyl-6-(trifluoromethyl)pyridine as starting material: 2-methyl-6-(trifluoromethyl)pyridine (100 mg, 0.620 mmol) was dissolved in carbon tetrachloride (CCl4, 3 mL), and N-bromosuccinimide (NBS, 110 mg, 0.620 mmol) and 1,1'-azobis(cyclohexanenitrile) (VAZO, 8 mg, 0.031 mmol). The reaction mixture was heated and stirred at 90 °C for 15 hours. After completion of the reaction, the reaction solution was filtered and the filtrate was concentrated under reduced pressure. The resulting residue was purified by medium pressure liquid chromatography (MPLC, hexane/ethyl acetate = 5:1) to afford 2-(bromomethyl)-6-(trifluoromethyl)pyridine as a white solid (42.5 mg, 28.4% yield).1H NMR (300 MHz, CDCl3) δ 4.60 (2H, s), 7.61 (1H, d), 7.68 (1H, d) , 7.90 (1H, t).