Preparation of 3,20-Disemicarbazone of 9α-Fluoro-11β,17α,21-Trihydroxy-16αMethyl-1,4-Pregnadiene-3,20-Dione: A mixture of 1.00 gram of 9α-fluoro11β,17α,21-trihydroxy-16α-methyl-1,4-pregnadiene-3,20-dione, 750 mg of semicarbazide base, 280 mg of semicarbazide hydrochloride in 20 ml of methanol and 10 ml of dimethylformamide is refluxed for 20 hours under nitrogen. The mixture is cooled to 20°C and 100 ml of water is added with stirring. The precipitated 3,20-disemicarbazone of 9α-fluoro-11β,17α,21trihydroxy-16α-methyl-1,4-pregnadiene-3,20-dione is filtered, washed with water, and dried in air; MP over 300°C.
Preparation of 9α-Fluoro-11β,21-Dihydroxy-16-Methyl-1,4,16-Pregnatriene3,20-Dione 21-Acetate: A solution of 500 mg of the 3,20-disemicarbazone of 9α-fluoro-11β,17α,21-trhydroxy-16α-methyl-1,4-pregnadiene-3,20-dione in 10 ml of acetic acid and 0.5 ml acetic anhydride is refluxed under nitrogen for one hour to produce the corresponding 3,20-disemicarbazone of 11β,21dihydroxy-16-methyl-1,4,16-pregnatriene-3,20-dione 21-acetate. The reaction mixture is cooled, 13 ml of water is added and the mixture heated on thesteam bath for 5 hours. It is then concentrated in vacuo nearly to dryness and water and chloroform added. The mixture is thoroughly extracted with chloroform, and the chloroform extract washed with excess aqueous potassium bicarbonate and saturated salt solution and dried over magnesium sulfate. Chromatography of the residue on neutral alumina and crystallization of pertinent benzene-chloroform fractions gives 9α-fluoro-11β,21-dihydroxy16-methyl-1,4,16-pregnatriene-3,20-dione 21-acetate; MP 228° to 233°C.
Preparation of 9α-Fluoro-11β,21-Dihydroxy-16β-Methyl-16α,17β-Oxido-1,4Pregnadiene-3,20-Dione 21-Acetate: To a stirred solution of 500 mg of 9αfluoro-11β,21-dihydroxy-16-methyl-1,4,16-pregnatriene-3,20-dione 21-acetate in 5 ml of benzene and 5 ml of chloroform are added 0.50 ml of t-butyl hydroperoxide and 0.1 ml of a 35% methanolic solution of benzyl-trimethyl ammonium hydroxide. After 18 hours at room temperature, water is added and the mixture thoroughly extracted with chloroform. The chloroform extract is washed with saturated aqueous sodium chloride and dried over magnesium sulfate. Evaporation of the solvent and crystallization of the residue from acetone-ether gives 9α-fluoro-11β,21-dihydroxy-16β-methyl-16α,17α-oxido1,4-pregnadiene-3,20-dione 21-acetate.
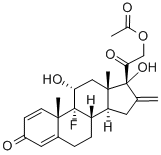
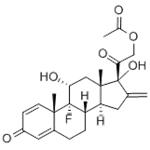
Preparation of 9α-Fluoro-11β,17α,21-Trihydroxy-16-Methylene-1,4Pregnadiene-3,20-Dione 21-Acetate: To a stirred solution of 600 mg of 9αfluoro-11β,21-dihydroxy-16β-methyl-16α,17α-oxido-1,4-pregnadiene-3,20dione21 acetate in 10 ml of acetic acid maintained at 10° to 15°C is added 3 ml of cold 10% hydrogen bromide in acetic acid. After 30 minutes the mixture is concentrated to dryness in vacuo (temperature 15°C) and the residue chromatographed on neutral alumina. Combination of pertinent benzenechloroform fractions and crystallization leads to the desired 9α-fluoro11β,17α,21-trihydroxy-16-methylene-1,4-pregnadiene-3,20-dione 21-acetate.