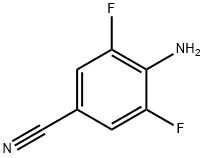
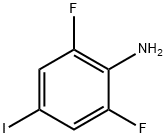
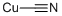General procedure for the synthesis of 4-amino-3,5-difluorobenzonitrile from 2,6-difluoro-4-iodoaniline and cuprous cyanide: a mixture of 2,6-difluoro-4-iodoaniline and cuprous cyanide in N,N-dimethylformamide (DMF) was heated and refluxed for 48 hours. Upon completion of the reaction, the mixture was filtered through a diatomaceous earth pad to remove insoluble impurities. The filtrate was transferred to a partition funnel and partitioned by adding dichloromethane and water. The extraction of the aqueous phase was repeated twice with dichloromethane. All organic phases were combined and washed with saturated brine and subsequently dried over anhydrous sodium sulfate. The desiccant was removed by filtration and the filtrate was concentrated under reduced pressure to give the crude product. The crude product was purified by column chromatography using hexane/ethyl acetate (4:1, v/v) as eluent, resulting in the target product 4-amino-3,5-difluorobenzonitrile (1.03 g, 66% yield) in yellow solid form. The product was characterized by 1H NMR (300 MHz, CDCl3): δ 7.15 (dd, 2H, J = 2.4, 6.0 Hz), 4.28 (br s, 2H).