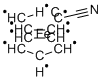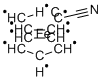Cyanoferrocene, [Fe(C5H5)(C6H4N)], forms sheets through hydrogen bonding. The N atom in the molecule accepts hydrogen bonds from C-H bonds in two other molecules, resulting in each molecule being hydrogen-bonded to four others. cyanoferrocene has occupied an important position since it can be converted to a structurally diverse set of ferrocenyl compounds that are attractive synthetic targets due to their chemical and biological properties.
The synthesis of cyanoferrocene involved treating 3.1g (0.01 mol) of iodoferrocene and 3.6g (0.04 mol) of copper(I) cyanide with 20 ml of pyridine, using the same procedure used for preparing chloroferrocene. The resulting crude product was purified by chromatography on Florisil, yielding 1.75 g of cyanoferrocene with a yield of 83%. The melting point of cyanoferrocene was 106-107°C (lit. 107-108°C). A by-product of unidentified yellow crystals was also obtained, weighing 0.15g with a melting point of 157-158°C.