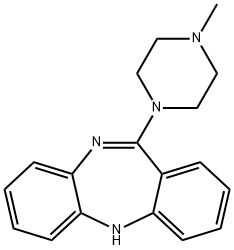
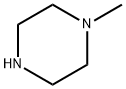
General procedure for the synthesis of 11-(4-methylpiperazin-1-yl)-5H-dibenzo[b,E][1,4]diazepin-11-one from N-methylpiperazine and 5,10-dihydro-11H-dibenzo[b,e][1,4]diazepin-11-one: 5,10-dihydro-11H-dibenzo[b,e][1,4]diazepin-11-one (105 mg , 0.5 mmol) was placed in a 50 mL round bottom flask. N,N-dimethylaniline (36.3 mg, 0.3 mmol) was added sequentially. An appropriate amount of super-dry trichlorophosphorus and a magnetic stirrer were added and the mixture was stirred under reflux conditions for 12 h at 130°C under nitrogen protection. Upon completion of the reaction, the excess phosphorous trichloride was removed by distillation. Subsequently, the reaction was terminated by adding an appropriate amount of N-methylpiperazine and continued stirring for 12 hours. The progress of the reaction was monitored by thin layer chromatography (TLC). The reaction mixture was extracted with ethyl acetate (3 x 70 mL) and washed sequentially with water. The organic layers were combined and dried by adding anhydrous Na2SO4. After filtration, the residue was concentrated. Purification by column chromatography (eluent: dichloromethane/methanol = 25:1) afforded 110 mg of the target product 11-(4-methylpiperazin-1-yl)-5H-dibenzo[b,E][1,4]diazepine in 75% yield.
![5,10-DIHYDRO-DIBENZO[B,E][1,4]DIAZEPIN-11-ONE](/CAS/GIF/5814-41-5.gif)


