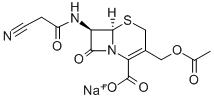
Cephacetrile was synthesized by Ciba-Geigy in 1970. It shows almost the same activity against gram-negative bacteria as cephalothin, but it has a higher activity against β-lactamaseproducing Escherichia coli. Like cephalothin, it is metabolized partially by deacetylation in vivo. Cephacetrile has been used to treat sepsis and abdominal and respiratory tract infections caused by Staphylococcus, Streptococcus, Escherichia coli, and Klebsiella pneumoniae, but it is gradually being replaced clinically by the newer and more active cephalosporins.
Celospor,Ciba Geigy,Switz.,1969
Cefacetrile Sodium is a cephalosporin antibiotic. Antibacterial.
ChEBI: Cefacetrile sodium is an organic molecular entity.
13.6 g (0.05 mol) of 7-aminocephalosporanic acid are taken up in a mixture
of 150 ml of methylene chloride and 19.5 ml of tributylamine (0.12 mol) and
at 0°C a solution of 8.4 g of cyanoacetylchloride (0.07 mol) in 100 ml of
methylene chloride is stirred in. The bath is then stirred for hour at 0°C
and for hour at 20°C, the reaction solution is evaporated under vacuum
and the residue taken up in 10% aqueous dipotassium hydrogenphosphate
solution. This aqueous phase is washed with ethyl acetate, acidified to pH 2.0
with concentrated hydrochloric acid and extracted with ethyl acetate.
After having been dried over sodium sulfate and evaporated under vacuum,
this extract gives as a solid residue 14.7 g of crude 7-cyanoacetylamino-cephalosporanic acid which is purified by chromatography on 30 times its own
weight of silica gel. The fractions eluted with chloroform plus acetone (7:3)
furnish a product which crystallizes from acetone plus ether in the form of
needles melting at 168° to 170°C with decomposition.
5.10 g (15 mmol) of 7-cyanoacetyl-aminocephalosporanic acid are suspended in 102 ml of distilled water and converted into the sodium salt by stirring in
dropwise 15 ml of N sodium hydroxide solution.