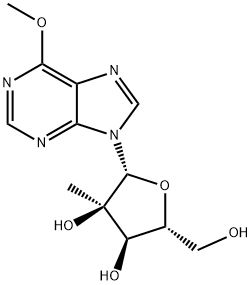
6-Methoxy-9-(2-C-methyl-beta-D-ribofuranosyl)purine is a potent antiviral compound used in the research of viral infections, particularly those caused by herpes viruses. It exhibits a high selectivity index and low toxicity towards host cells, making it an effective option for suppressing viral replication. This nucleoside analogue inhibits the viral DNA polymerase, thus preventing viral DNA research and development and subsequent viral replication.
2′-C-Methyl-6-O-methylinosine is a hypoxanthine analog. Hypoxanthine is a kind of purine base mainly present in muscle tissue. And it is a metabolite produced by purine oxidase acting on xanthine. Hypoxanthine has typical anti-inflammatory effects and is a potential endogenous poly(ADP-ribose) polymerase (PARP) inhibitor. It is cytoprotective by inhibiting PAPR activity, inhibiting peroxynitrite-induced mitochondrial depolarization and secondary superoxide production. Hypoxanthine can also be used as an indicator of hypoxia[1][2].