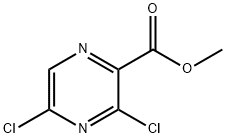
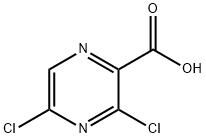
At 23 °C, 3,5-dichloropyrazine-2-carboxylic acid (2.68 g, 13.9 mmol) and sodium bicarbonate (1.4 g, 16.6 mmol) were dissolved in DMF (20 mL), followed by addition of iodomethane (5.21 mL, 83 mmol). After completion of the reaction, the reaction mixture was diluted with 10% aqueous citric acid and extracted with EtOAc. The organic layers were combined, washed sequentially with water and brine, dried over anhydrous MgSO4 and concentrated under reduced pressure to afford the brown solid product methyl 3,5-dichloropyrazine-2-carboxylate (2.83 g, 13.6 mmol, 98% yield). The structure of the product was confirmed by 1H NMR (400 MHz, DMSO-d6): δ 3.95 (s, 3H), 8.94 (s, 1H).