Standard
In the analysis work, it often demanded to apply high-quality compound with the exact content being accurate in order to be compared as the detection control or used to calibrate the instrument, the compound is known as standard sample.
Standard refers to the standard substance used for measuring the potency of biological products. When the production department submits to a higher authority for approval of the quality standard of the biological product, they should also provide the standard sample (indicated in unit (μ)) used for measuring the potency of the tested product.
According to different application, the verification products can be divided into biological standard for the testing of biological products, endocrine drugs, antibiotics and other bioassay and chemical standards including:
1 chemical standards used for calibration of the instrument;
2 chemical reference standards used for calibration of the ultraviolet or infrared spectrum of the drugs;
3 control standards used for the purity test of drugs as well as the impurity control or as the control standards for content determination of the medicine.
In the page 67 of the appendix in the People's Republic of China Pharmacopoeia 1990 1st edition, we can find hundreds kinds of standards including bornyl ester acetate, eugenol and panaxadiol as well as 39 kinds of control medicine for the purify inspection of traditional Chinese medicine, impurity control or the content determination.
International standards
This refers to standard sample, with the potency units of the existing international standard sample potency units as baseline, undergoing further calibration of its potency units on the basis of a wide range of international cooperation. Its potency is expressed in 1U. For a certain product, for the establishment of the first international standard sample, the determination of its potency units should be consulted and decided by the Committee of Experts before the announcement. The international standard is a standard reference substance for worldwide preparation of national standards, so that the international standards can achieve unity.
Reagents and standards
All reagents in the food analysis laboratories should be clearly labeled to indicate the following: 1 name; 2 the date of preparation; 3 Signature from the people in charge; 4 if necessary, we need to add content such as safety and validity and so on.
Most food analysis should use standard sample as reference for qualitative and quantitative determination. The standard sample should be classified according to the following classification methods: 1 international standard; 2 national standards; 3 internal standard; 4 other standards.
Receptacle for reference standards should be clearly affixed to the label, to indicate the following: 1 name; 2 standard document number; 3 Opening times; 4 Validity; 5 Signature.
In addition, the laboratory should keep all the necessary specific files for all the necessary reference standards.
Capacity analysis standard solution should also be strictly managed. It should be clearly posted on all the standard solution containers with labels to indicate: 1 name; 2 concentration; 3 formulation date; 4 No of formulated procedures; 5 storage conditions; 6 validity; 7 Signature from people in charge.
- Structure:
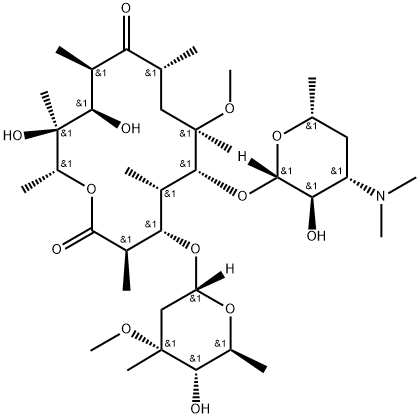
- Chemical Name:Clarthromycin EP Impurity B
- CAS:299409-85-1
- MF:C37H67NO13
- Structure:
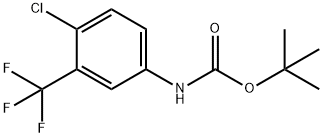
- Chemical Name:(4-Chloro-3-trifluoromethyl-phenyl)-carbamic acid tert-butyl ester
- CAS:352277-93-1
- MF:C12H13ClF3NO2
- Chemical Name:Brexpiprazole Impurity 15
- CAS:
- MF:
- Structure:
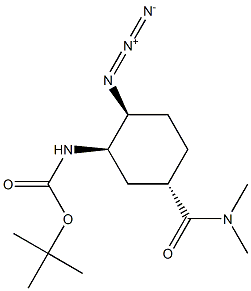
- Chemical Name:Edoxaban Impurity 16
- CAS:480450-69-9
- MF:C14H25N5O3
- Chemical Name:Rivaroxaban Impurity 24
- CAS:
- MF:
- Structure:
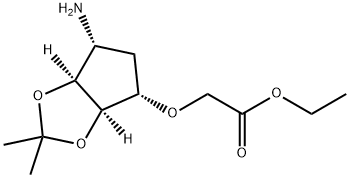
- Chemical Name:Ticagrelor
- CAS:1265919-24-1
- MF:C12H21NO5
- Structure:
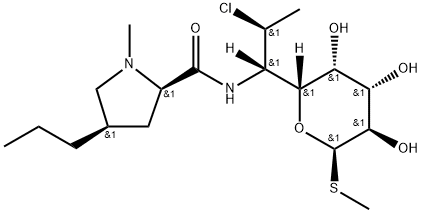
- Chemical Name:Clindamycin (2R-cis)-Diastereomer
- CAS:1440605-46-8
- MF:C18H33ClN2O5S
- Structure:
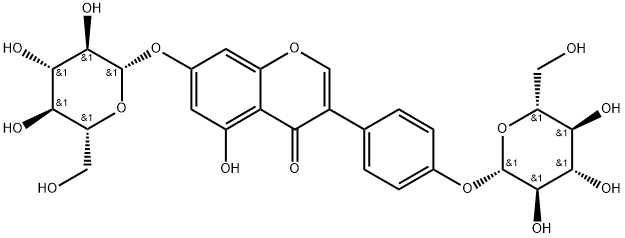
- Chemical Name:Genistein 7,4'-di-O-β-D-glucopyranoside
- CAS:36190-98-4
- MF:C27H30O15
- Chemical Name:Aprepitant Impurity 3
- CAS:
- MF:
- Chemical Name:Cabozantinib Impurity F
- CAS:
- MF:
- Structure:

- Chemical Name:Cefmetazole sodium Impurity 7
- CAS:60638-57-5
- MF:C11H14N6O4S2
- Chemical Name:Tadalafil EP Impurity I
- CAS:
- MF:
- Structure:
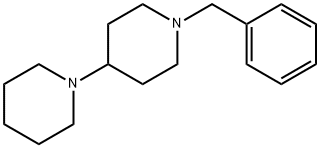
- Chemical Name:1,4'-Bipiperidine, 1'-(phenylmethyl)-
- CAS:116269-53-5
- MF:C17H26N2
- Structure:
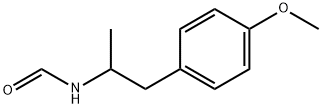
- Chemical Name:[2-(4-Methoxyphenyl)-1-methylethyl]-formamide
- CAS:126002-14-0
- MF:C11H15NO2
- Structure:
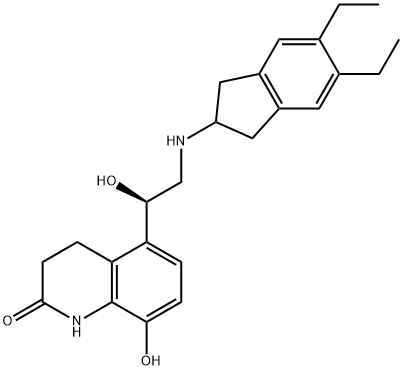
- Chemical Name:Indacaterol Impurity 7
- CAS:1403389-05-8
- MF:C24H30N2O3
- Structure:
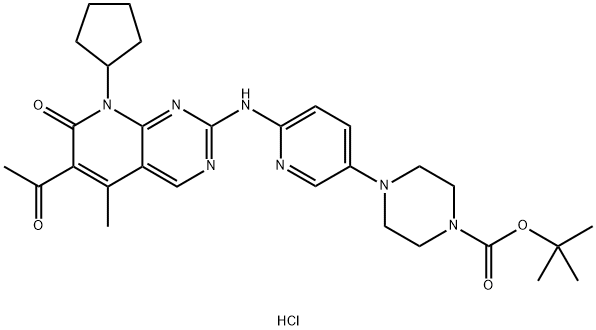
- Chemical Name:tert-butyl 4-(6-((6-acetyl-8-cyclopentyl-5-methyl-7-oxo-7,8-dihydropyrido[2,3-d]pyrimidin-2-yl)amino)pyridin-3-yl)piperazine-1-carboxylate
- CAS:1883672-48-7
- MF:C29H38ClN7O4
- Structure:

- Chemical Name:1-cyclopropyl-7-fluoro-6,8-dimethoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- CAS:
- MF:C15H14FNO5
- Structure:
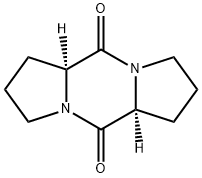
- Chemical Name:cis-cyclo(Pro-Pro)
- CAS:19943-27-2
- MF:C10H14N2O2
- Structure:
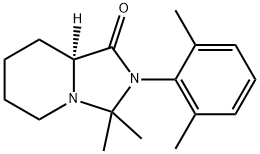
- Chemical Name:Ropivacaine-cycle
- CAS:1945965-95-6
- MF:C17H24N2O
- Structure:
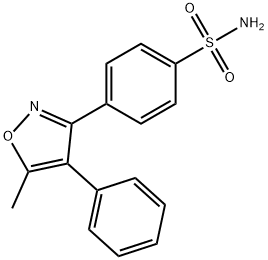
- Chemical Name:4-(5-methyl-4-phenylisoxazol-3-yl)benzenesulfonamide
- CAS:181696-12-8
- MF:C16H14N2O3S
- Structure:
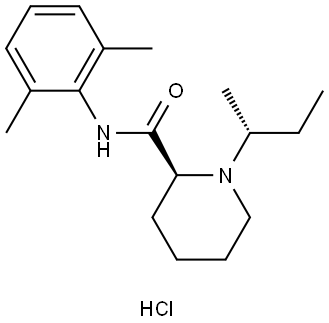
- Chemical Name:Bupivacaine Impurity
- CAS:2125995-27-7
- MF:C18H28N2O.ClH
- Structure:
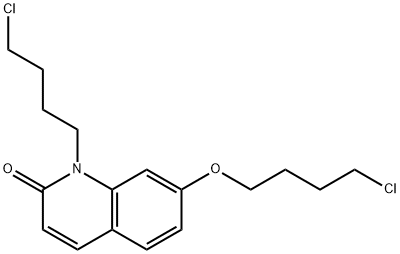
- Chemical Name:Brexpiprazole Impurity
- CAS:2059954-32-2
- MF:C17H21Cl2NO2
- Structure:
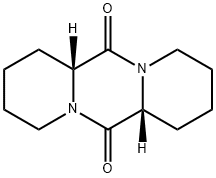
- Chemical Name:BDJS-SH 2
- CAS:36585-31-6
- MF:C12H18N2O2
- Chemical Name:Acyclovir Impurity Q
- CAS:
- MF:
- Structure:
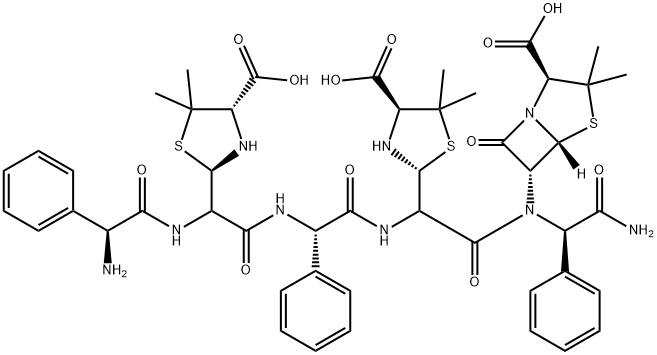
- Chemical Name:Ampicillin EP Impurity M
- CAS:114977-84-3
- MF:C48H57N9O12S3
- Structure:
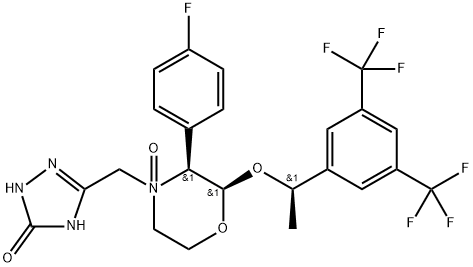
- Chemical Name:Aprepitant Impurity 12
- CAS:172673-23-3
- MF:C23H21F7N4O4
- Chemical Name:Aprepitant Impurity 18
- CAS:
- MF:
- Chemical Name:Blonanserin impurity D
- CAS:
- MF:
- Chemical Name:Captopril impurity 3
- CAS:
- MF:
- Chemical Name:Empagliflozin Impurity 18
- CAS:
- MF:
- Chemical Name:Empagliflozin Impurity 24
- CAS:
- MF:
- Chemical Name:FeBuxostat impurity 31
- CAS:
- MF:
- Structure:
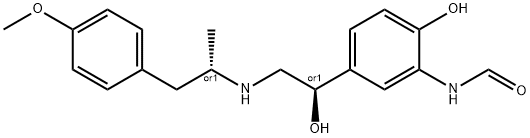
- Chemical Name:Formoterol Fumarate Dihydrate EP Impurity I
- CAS:532414-36-1
- MF:C19H24N2O4
- Chemical Name:Fosaprepitant Impurity 3
- CAS:
- MF:
- Structure:
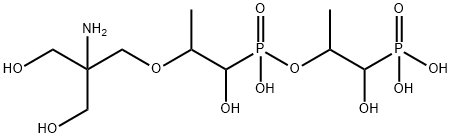
- Chemical Name:Fosfomycin Trometamol EP Impurity D
- CAS:1262243-12-8
- MF:C10H25NO11P2
- Chemical Name:Isavuconazole Impurity 17
- CAS:
- MF:
- Chemical Name:Isavuconazole Impurity 5
- CAS:
- MF:
- Chemical Name:Moxifloxacin Impurity 21
- CAS:
- MF:
- Structure:

- Chemical Name:Mupirocin EP Impurity A
- CAS:
- MF:C26H44O9
- Structure:
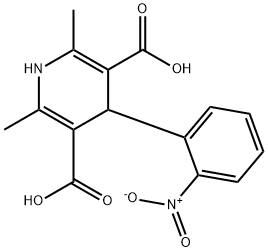
- Chemical Name:Nifendipine Impurity G
- CAS:74378-10-2
- MF:C15H14N2O6
- Chemical Name:Olaparib Impurity 5
- CAS:
- MF:
- Chemical Name:Plerixafor Impurity 5
- CAS:
- MF:
- Chemical Name:Posaconazole Impurity 30
- CAS:
- MF:
- Structure:

- Chemical Name:Posaconazole Impurity 35
- CAS:1370190-85-4
- MF:C14H13F2N3O
- Structure:
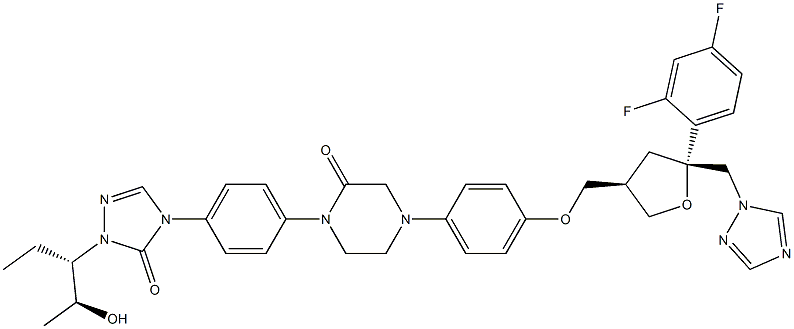
- Chemical Name:Posaconazole Impurity 51
- CAS:357189-94-7
- MF:C37H40F2N8O5
- Chemical Name:Posaconazole Impurity 59
- CAS:
- MF:
- Structure:
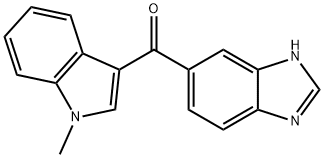
- Chemical Name:Ramosetron Impurity 13
- CAS:183613-77-6
- MF:C17H13N3O
- Structure:
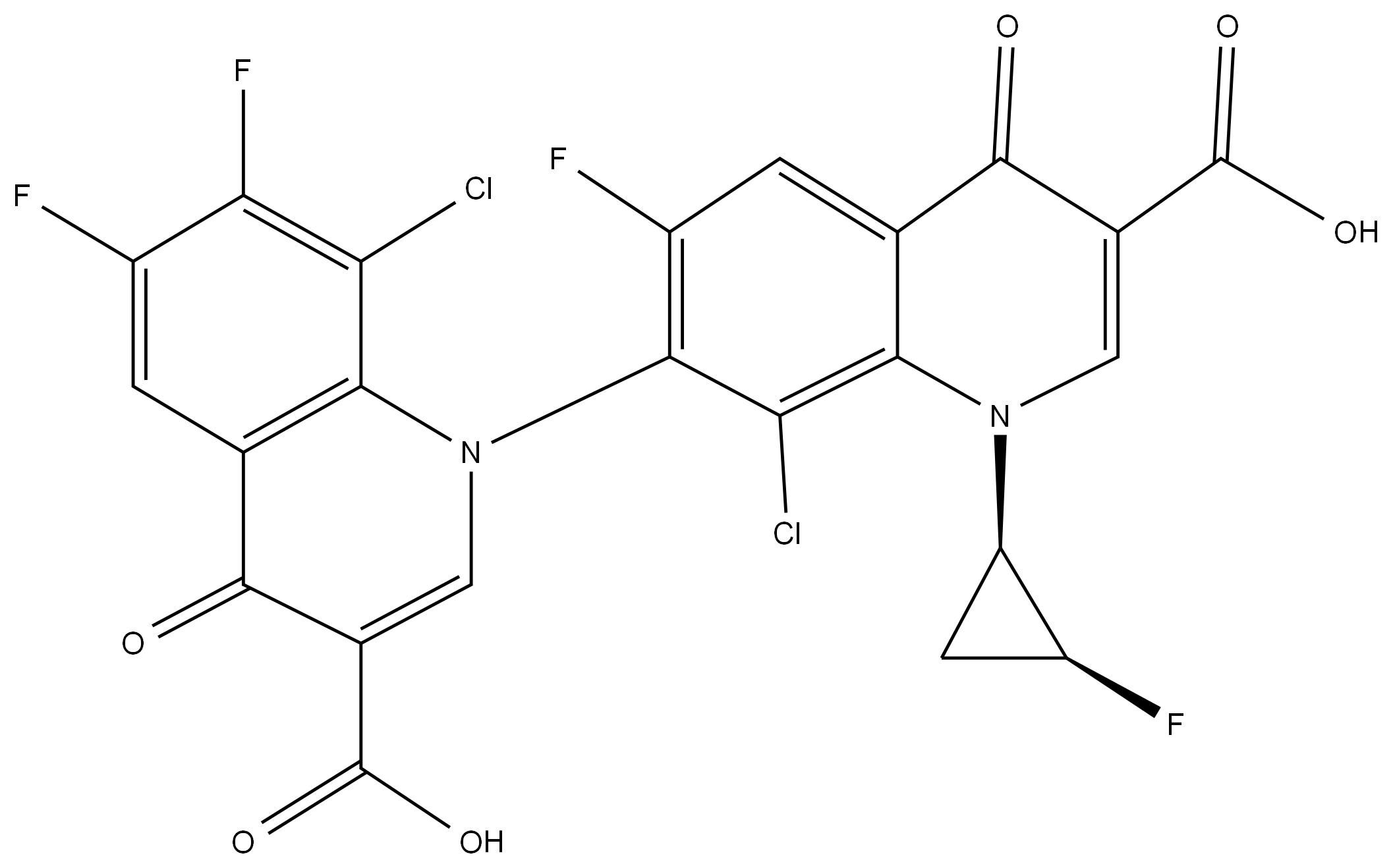
- Chemical Name:Sitafloxacin Impurity 2
- CAS:
- MF:C23H10Cl2F4N2O6
- Structure:
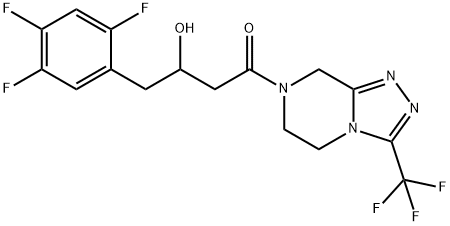
- Chemical Name:Sitagliptin Impurity 10
- CAS:1253056-01-7
- MF:C16H14F6N4O2
- Chemical Name:SofosBuvir Impurity 18
- CAS:
- MF:
- Chemical Name:sofosBuvir Impurity 35
- CAS:
- MF:
- Chemical Name:SofosBuvir Impurity 51
- CAS:
- MF:
- Chemical Name:SofosBuvir Impurity 58
- CAS:
- MF:
- Chemical Name:Sulbactam EP Impurity G
- CAS:
- MF:
- Structure:
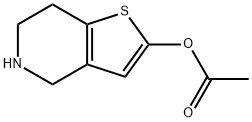
- Chemical Name:4,5,6,7-Tetrahydro-thieno[3,2-c]pyridin-2-ol 2-acetate
- CAS:1151904-85-6
- MF:C9H11NO2S
- Structure:
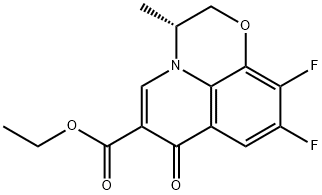
- Chemical Name:ethyl (R)-9,10-difluoro-3-methyl-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylate
- CAS:110548-06-6
- MF:C15H13F2NO4
- Structure:
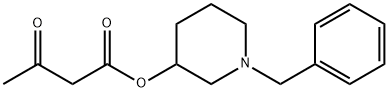
- Chemical Name:Butanoic acid, 3-oxo-, 1-(phenylmethyl)-3-piperidinyl ester
- CAS:85387-34-4
- MF:C16H21NO3
- Chemical Name:Ticagrelor impurity 21
- CAS:
- MF:
- Structure:
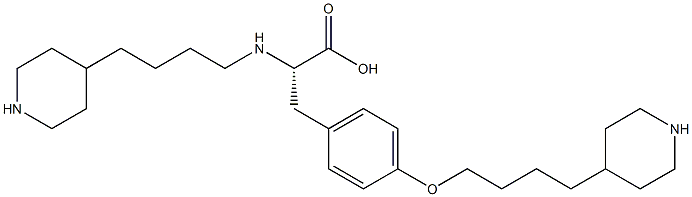
- Chemical Name:Tirofiban Impurity 13
- CAS:
- MF:C27H45N3O3
- Chemical Name:Tofacitinib impurity U
- CAS:
- MF:
- Structure:
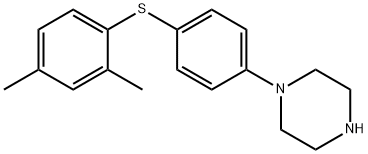
- Chemical Name:Vortioxetine impurity S
- CAS:1815608-51-5
- MF:C18H22N2S
- Structure:
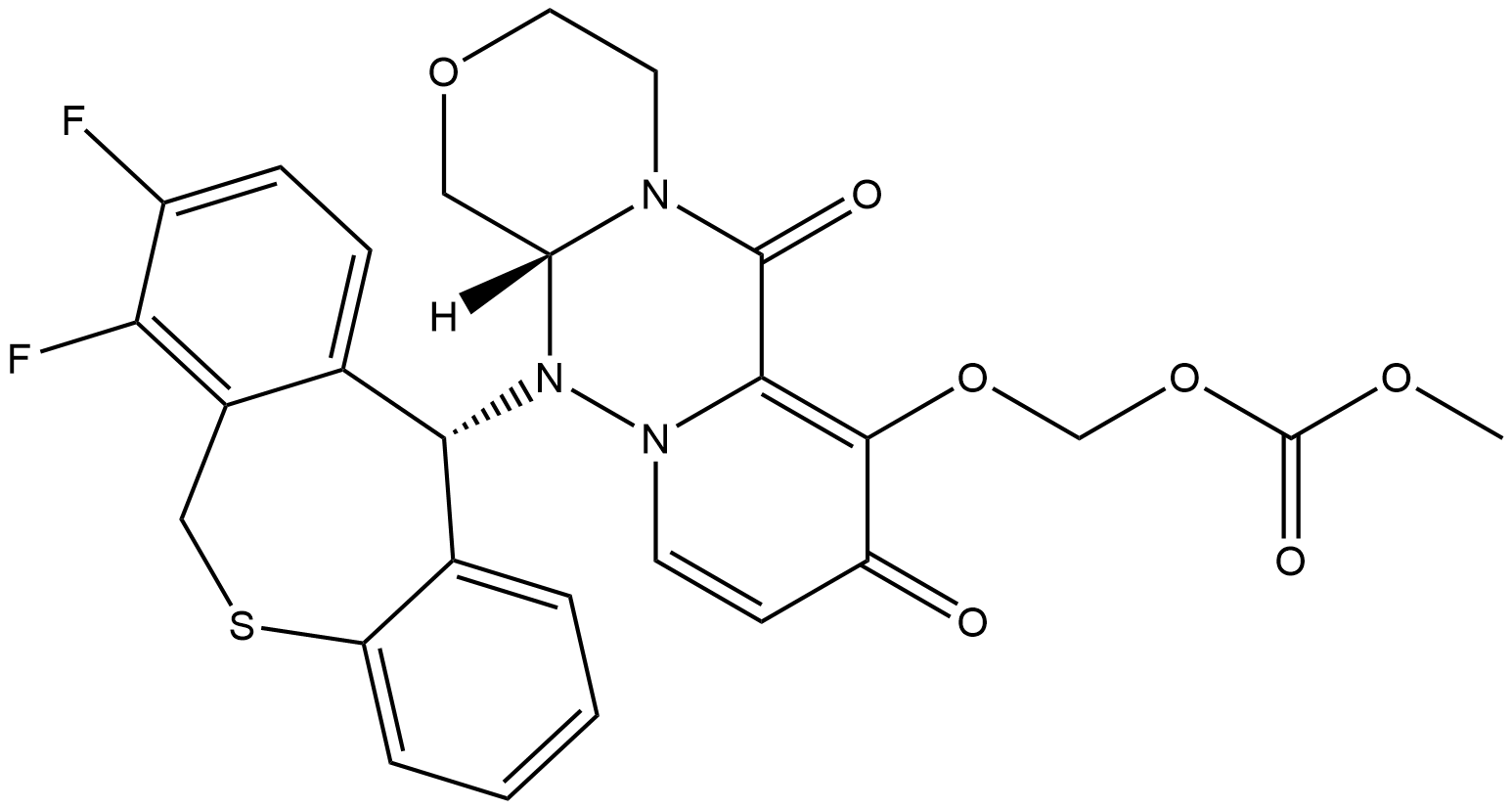
- Chemical Name:Baloxavir marboxil
- CAS:1985606-14-1
- MF:C27H23F2N3O7S
- Structure:
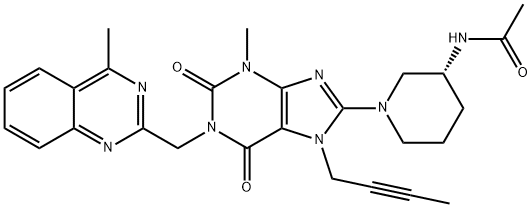
- Chemical Name:Linagliptin N-Acetyl Impurity
- CAS:1803079-49-3
- MF:C27H30N8O3
- Structure:
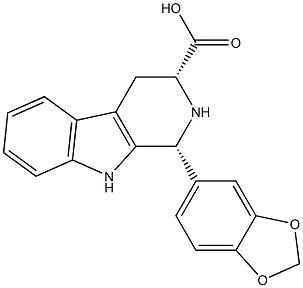
- Chemical Name:(1R,3R)-1-(benzo[d][1,3]dioxol-5-yl)-2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole-3-carboxylic acid
- CAS:474668-76-3
- MF:C19H16N2O4
- Structure:
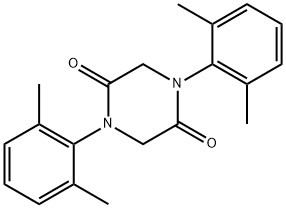
- Chemical Name:Lidocaine Impurity
- CAS:70336-49-1
- MF:C20H22N2O2
- Structure:
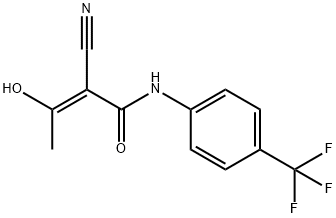
- Chemical Name:(E)-2-cyano-3-hydroxy-N-[4-(trifluoromethyl)phenyl]but-2-enamide
- CAS:214782-56-6
- MF:C12H9F3N2O2
- Structure:
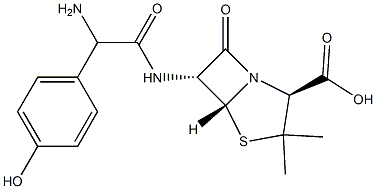
- Chemical Name:Amoxicillin Impurity O
- CAS:
- MF:C16H19N3O5S
- Chemical Name:Ampicillin Impurity R
- CAS:
- MF:
- Chemical Name:Spectinomycin Impurity C
- CAS:
- MF:
- Chemical Name:Amifostine Impurity 2
- CAS:
- MF:
- Structure:
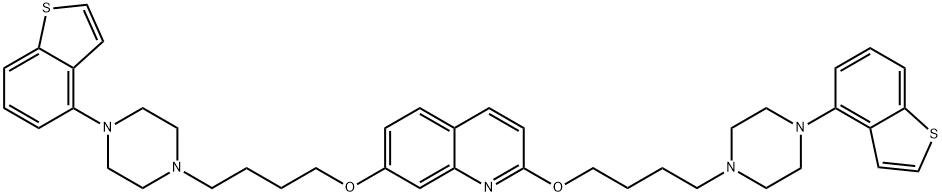
- Chemical Name:Brexpiprazole Impurity
- CAS:2138169-93-2
- MF:C41H47N5O2S2
- Chemical Name:Cefoxitin impurity EF
- CAS:
- MF:
- Structure:
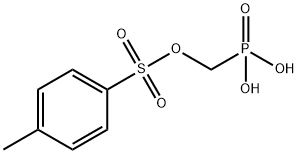
- Chemical Name:Tenofovir Impurity 38
- CAS:161760-09-4
- MF:C8H11O6PS
- Structure:
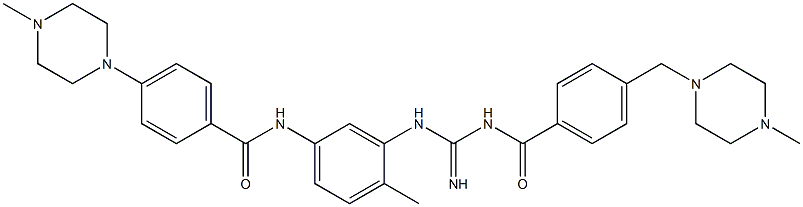
- Chemical Name:N-(4-methyl-3-(3-(4-((4-methylpiperazin-1-yl)methyl)benzoyl)guanidino)phenyl)-4-(4-methylpiperazin-1-yl)benzamide
- CAS:
- MF:C33H42N8O2
- Structure:
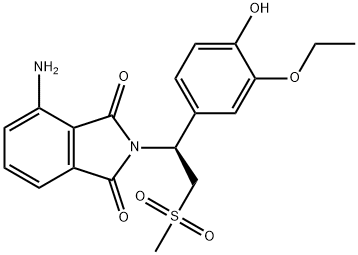
- Chemical Name:Apremilast
- CAS:1384440-16-7
- MF:C19H20N2O6S
- Structure:
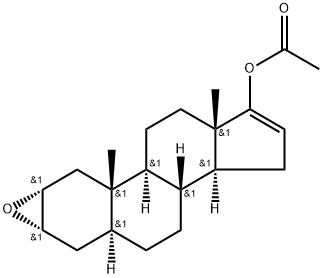
- Chemical Name:XMUJPIDSOJTMMS-VTBMCCKRSA-N
- CAS:212505-49-2
- MF:C21H30O3
- Structure:
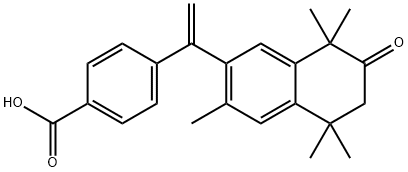
- Chemical Name:XLMHAABXFLIUNQ-UHFFFAOYSA-N
- CAS:368451-15-4
- MF:C24H26O3
- Structure:
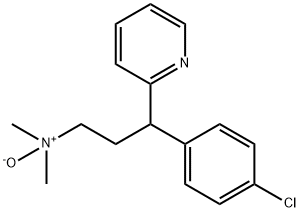
- Chemical Name:NMEICKDXQPVPKK-UHFFFAOYSA-N
- CAS:120244-82-8
- MF:C16H19ClN2O
- Structure:
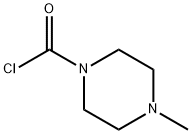
- Chemical Name:4-Methylpiperazine-1-carbonyl chloride
- CAS:39539-66-7
- MF:C6H11ClN2O
- Structure:
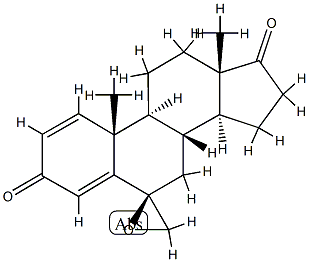
- Chemical Name:Epoxy Exemestane (6-Beta Isomer)
- CAS:152764-31-3
- MF:C20H24O3
- Structure:
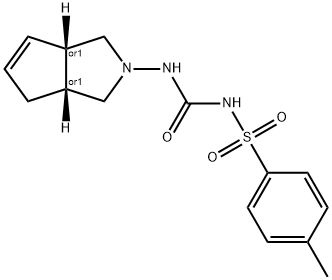
- Chemical Name:AFTMPFJWAIDLME-QWHCGFSZSA-N
- CAS:1808087-53-7
- MF:C15H19N3O3S
- Structure:
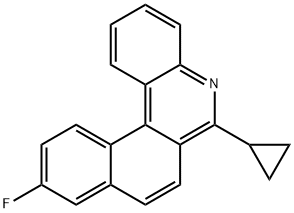
- Chemical Name:6-Cyclopropyl-10-fluorobenzo[k]phenanthridine
- CAS:1187966-95-5
- MF:C20H14FN
- Structure:
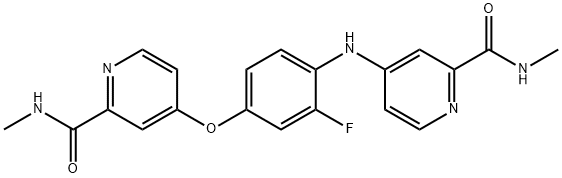
- Chemical Name:4-[2-fluoro-4-[2-(methylcarbamoyl)pyridin-4-yl]oxyanilino]-N-methylpyridine-2-carboxamide
- CAS:1855006-12-0
- MF:C20H18FN5O3
- Structure:
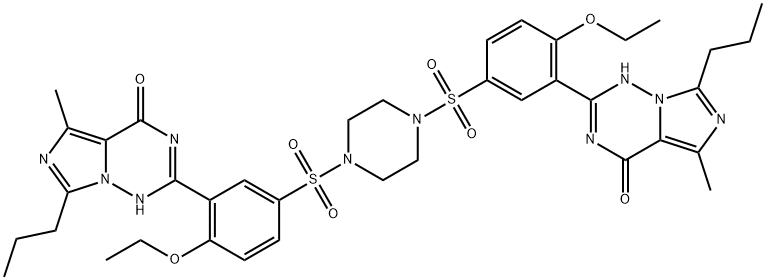
- Chemical Name:2,2'-((piperazine-1,4-disulfonyl)bis(2-ethoxy-5,1-phenylene))bis(5-methyl-7-propylimidazo[5,1-f][1,2,4]triazin-4(3H)-one)
- CAS:1255919-03-9
- MF:C38H46N10O8S2
- Chemical Name:Bortezomib Impurity P
- CAS:
- MF:
- Chemical Name:Esmolol impurity A
- CAS:
- MF:
- Structure:
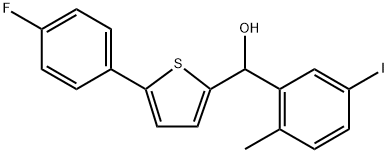
- Chemical Name:(5-(4-fluorophenyl)thiophen-2-yl)(5-iodo-2-methylphenyl)methanol
- CAS:1818268-45-9
- MF:C18H14FIOS
- Structure:
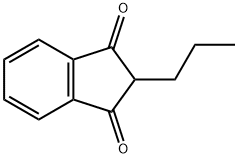
- Chemical Name:2-propyl-1H-indene-1,3(2H)-dione
- CAS:14570-43-5
- MF:C12H12O2
- Structure:
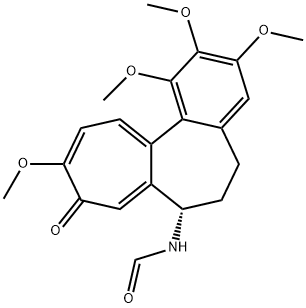
- Chemical Name:GLORIOSINE
- CAS:7411-12-3
- MF:C21H23NO6
- Structure:

- Chemical Name:N-METHYL-3-CHLORO-3-PHENYL PROPYLAMINE HCL
- CAS:128036-32-8
- MF:C10H15Cl2N
- Structure:
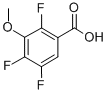
- Chemical Name:2,4,5-TRIFLUORO-3-METHOXYBENZOIC ACID
- CAS:11281-65-5
- MF:C8H5F3O3
- Structure:
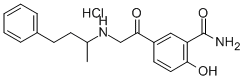
- Chemical Name:Labetalone hydrochloride
- CAS:96441-14-4
- MF:C19H22N2O3.ClH
- Structure:
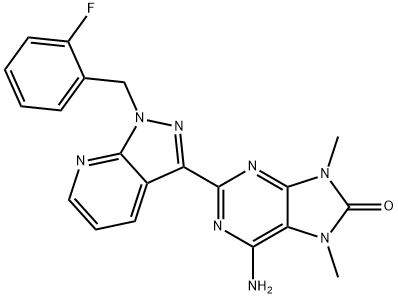
- Chemical Name:6-amino-2-(1-(2-fluorobenzyl)-1H-pyrazolo[3,4-b]pyridin-3-yl)- 7,9-dimethyl-7,9-dihydro-8H-purin-8-one
- CAS:1361569-23-4
- MF:C20H17FN8O
- Structure:
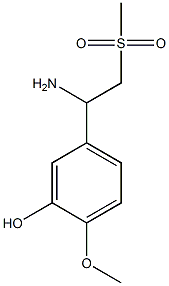
- Chemical Name:5-(1-amino-2-(methylsulfonyl)ethyl)-2-methoxyphenol
- CAS:2169153-70-0
- MF:C10H15NO4S
- Chemical Name:Bendamustine Impurity 14
- CAS:
- MF:
- Chemical Name:Canagliflozin Impurity 28
- CAS:
- MF:
- Structure:

- Chemical Name:Edoxaban Impurity 24 (1S,2S,4R)
- CAS:2024614-21-7
- MF:C21H30ClN5O5
- Structure:
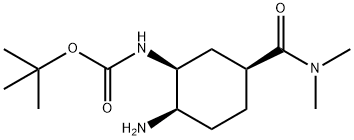
- Chemical Name:Edoxaban Impurity 39
- CAS:2081883-55-6
- MF:C14H27N3O3
- Structure:
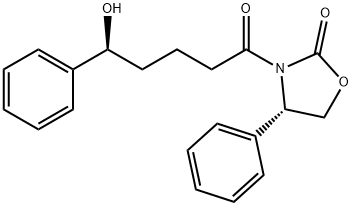
- Chemical Name:Ezetimibe Impurity 34
- CAS:2280081-69-6
- MF:C20H21NO4
- Structure:
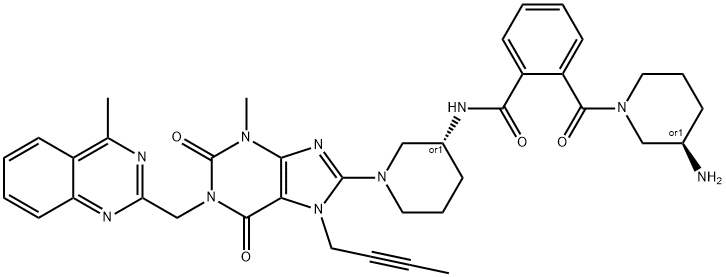
- Chemical Name:Linagliptin Impurity 33
- CAS:2443735-53-1
- MF:C38H42N10O4