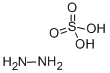Hydrazine sulfate is salt which generated by hydrazine and sulfuric acid, pure product is colorless scaly crystal or rhombic. The molecular weight is 130.12. Formula is N2H4·H2SO4. Melting point is 254℃, when continues heating, it can decompose. The relative density is 1.37. It is slightly soluble in water, soluble in hot water (at 20℃ 2.87, at 25℃ 3.41, at 30℃ 3.89, at 40℃ 4.16, at 50 ℃ 7.0, at 60℃ 9.07, at 80℃ 14.4), aqueous solution is acidic , it is insoluble in alcohol and ether. It is very stable in air. It is susceptible to alkali and oxidizing agents, it is incompatible with bases, oxidants. It has strong reduction. Rat oral LD50601mg/kg, it is toxic, carcinogenic. The main purpose of hydrazine sulfate is used for determination the weight of nickel, cobalt, cadmium, and the purification of rare metals, the separation of tellurium and polonium, as well as the precipitation of chlorate, hypochlorite and carboxyl compound, it is also used for manufacturing isoniazid , nitrofurazone, 100 Health hydrazine, anhydrous hydrazine, etc., it is also used as reducing agent, insecticides and sterilization agents. Hydrazine sulfate is nutritional supplement in United States for fighting against anorexia, weight loss and other symptoms patients which caused by cancer.
Laboratory method for preparing hydrazine sulfate
1. Sodium hypochlorite is prepared by chlorine and sodium hydroxid, then reacts with ammonia to obtain hydrazine hydrate, and finally to obtain hydrazine by dehydration, and then reacts with sulfuric acid to obtain hydrazine sulfate products. Related chemical reaction equation is as follows:
2NH3 + NaClO → NH2Cl + NaOH
NH2Cl + NaOH + NH3 → N2H4 + NaCl + H2O
N2H4 + H2SO4 → N2H4 · H2SO4
2. When ammonia reacts with water, ammonium hydroxide can be obtained, and then by synthesis, hydrazine solution can be obtained, by evaporation, condensation, crystallization, and finally reacts with sulfuric acid to obtain hydrazine sulfate products.