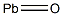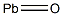Hazard statements
H341:Suspected of causing genetic defects
H350:May cause cancer
H360:May damage fertility or the unborn child
H362:May cause harm to breast-fed children
H370:Causes damage to organs
H373:May cause damage to organs through prolonged or repeated exposure
H410:Very toxic to aquatic life with long lasting effects
Precautionary statements
P201:Obtain special instructions before use.
P202:Do not handle until all safety precautions have been read and understood.
P260:Do not breathe dust/fume/gas/mist/vapours/spray.
P263:Avoid contact during pregnancy/while nursing.
P264:Wash hands thoroughly after handling.
P264:Wash skin thouroughly after handling.
P270:Do not eat, drink or smoke when using this product.
P271:Use only outdoors or in a well-ventilated area.
P273:Avoid release to the environment.
P280:Wear protective gloves/protective clothing/eye protection/face protection.
P391:Collect spillage. Hazardous to the aquatic environment
P405:Store locked up.
P501:Dispose of contents/container to..…