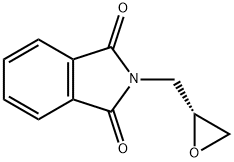
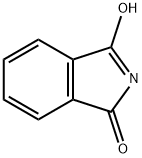
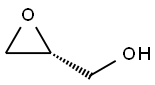
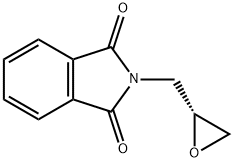
Chemical Properties
White solid
Uses
(
R)-(-)-
N-(2,3-Epoxypropyl)phthalimide may be used in the preparation of the following potential chiral solvating agents:
- 2-[(2R)-2-hydroxy-3-[[(1R)-2-hydroxy-1-phenyl-ethyl]amino]propyl] isoindoline-1,3-dione
- 2-[(2R)-2-hydroxy-3-[[(1R,2S)-2-hydroxyindan-1-yl]amino]propyl]isoindoline-1,3-dione
- 2-[(2R)-2-hydroxy-3-[[(1R)-1-(hydroxymethyl)-propyl]amino]propyl] isoindoline-1,3-dione
- 2-[(2R)-2-hydroxy-3-[[(1S)-1-phenylethyl]-amino]propyl]isoindoline-1,3-dione
It may also be used in the synthesis of 2-((5
R)-2-oxo-5-oxazolidinyl) methyl)-1
H-isoindole-1,3 (2
H)-dione.
Uses
(R)-N-Glycidylphthalimide is a reagent used for the simultaneous preparation of amino alcohol solvating agents as well as preparation of orally available anticoagulants.
Synthesis
General procedure for the synthesis of (R)-N-(2,3-epoxypropyl)phthalimide from 3-hydroxy-1H-isoindol-1-one and (R)-epoxyethylene-2-methanol: To a solution of tetrahydrofuran (THF, 220 mL) containing phthalimide (6.6 g, 45.0 mmol), triphenylphosphine (PPh3, 17.7 g, 67.5 mmol), diethyl azodicarboxylate (DEAD, 11.7 mL, 67.5 mmol), and (R)-(+)-condensate were sequentially added. 67.5 mmol), diethyl azodicarboxylate (DEAD, 11.7 mL, 67.5 mmol) and (R)-(+)-glycidol (4.0 g, 54.0 mmol). The reaction mixture was stirred at room temperature overnight. Upon completion of the reaction, the THF solvent was removed by distillation under reduced pressure and the residue was purified by silica gel fast column chromatography (Biotage system, eluent: 5%-20% ethyl acetate/hexane) to afford the target product (R)-N-(2,3-epoxypropyl)phthalimide (5.8 g, 75% yield).
References
[1] Tetrahedron Asymmetry, 1996, vol. 7, # 6, p. 1641 - 1648
[2] Patent: WO2006/29210, 2006, A2. Location in patent: Page/Page column 112-113
[3] Heterocycles, 2006, vol. 67, # 2, p. 561 - 566
[4] Tetrahedron, 2004, vol. 60, # 35, p. 7679 - 7692
[5] Patent: US2006/14701, 2006, A1. Location in patent: Page/Page column 23