Synthesis
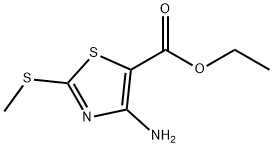
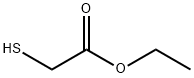
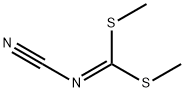
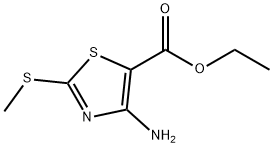
Synthesis of ethyl 4-amino-2-(methylthio)thiazole-5-carboxylate: ethyl 2-mercaptoacetate (50 g, 0.416 mol) was dissolved in 500 ml of dimethylformamide (DMF), followed by the addition of N-cyanoimino-S,S-dimethyl-dithiocarbonate (67 g, 0.416 mol) and diisopropylamine (112 ml, 0.624 mol). The reaction mixture was heated and stirred at 100 °C for 5 hours. After completion of the reaction, the mixture was poured into 500 ml of saturated ammonium chloride solution and extracted with 500 ml of ethyl acetate. The organic layer was dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The resulting solid was washed with hexane to give ethyl 2-methylthio-4-aminothiazole-5-carboxylate (90 g, 99% yield). The product was characterized by 1H-NMR (400 MHz, CDCl3): δ 5.84 (broad single peak, 2H), 4.26 (quadruple peak, J=7.2 Hz, 2H), 2.63 (single peak, 3H), 1.32 (triple peak, J=7.2 Hz, 3H); LC-MS showed the molecular ion peak m/z 219 (MH+).
References
[1] Patent: US2013/72482, 2013, A1. Location in patent: Paragraph 0315-0317
[2] Patent: WO2015/89327, 2015, A1. Location in patent: Paragraph 0468
[3] Tetrahedron Letters, 1966, # 17, p. 1885 - 1889
[4] Patent: WO2015/193506, 2015, A1. Location in patent: Page/Page column 57-58