ChemicalBook > CAS DataBase List > MEPHOBARBITAL
MEPHOBARBITAL
MEPHOBARBITAL
- CAS No.115-38-8
- Chemical Name:MEPHOBARBITAL
- CBNumber:CB2207809
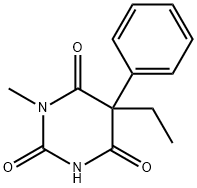
- Molecular Formula:C13H14N2O3
- Formula Weight:246.26
- MOL File:115-38-8.mol
MEPHOBARBITAL Property
- Melting point 1760C
- Boiling point 389.26°C (rough estimate)
- Density 1.1855 (rough estimate)
- refractive index 1.6450 (estimate)
- Flash point 11 °C
- storage temp. Controlled Substance, -20°C Freezer
- solubility Practically insoluble in water, very slightly soluble in ethanol (96 per cent).
- pka 7.99±0.10(Predicted)
- form Solid
- color White to Off-White
- Water Solubility 0.15g/L(20 ºC)
- CAS DataBase Reference 115-38-8
- EWG's Food Scores 1
- FDA UNII 5NC67NU76B
- ATC code N03AA01
- EPA Substance Registry System 2,4,6(1H,3H,5H)-Pyrimidinetrione, 5-ethyl-1-methyl-5-phenyl- (115-38-8)
Safety
- Hazard Codes :Xn,T,F
- Risk Statements :22-39/23/24/25-23/24/25-11
- Safety Statements :36/37/39-45-36/37-16-7
- RIDADR :3249
- HazardClass :6.1(b)
- PackingGroup :III
- HS Code :2933530000
- Hazardous Substances Data :115-38-8(Hazardous Substances Data)
- Toxicity :cat,LD50,oral,230mg/kg (230mg/kg),Drugs in Japan Vol. 6, Pg. 851, 1982.
-
Symbol(GHS)



- Signal wordDanger
- Hazard statements H225-H301+H311+H331-H370
- Precautionary statements P210-P280-P301+P310+P330-P302+P352+P312-P304+P340+P311
MEPHOBARBITAL Price
More Price(1)
- Brand: Sigma-Aldrich(India)
- Product number: M-193
- Product name : Methylphenobarbital
- Purity: 1.0?mg/mL in methanol, certified reference material, Cerilliant?
- Packaging: 1ML
- Price: ₹20388.9
- Updated: 2022/06/14
- Buy: Buy
MEPHOBARBITAL Chemical Properties,Usage,Production
- Chemical Properties White Crystalline Solid
- Originator Isonal, Roussel
- Uses Controlled substance (depressant). Anticonvulsant; sedative; hypnotic
- Definition ChEBI: A member of the class of barbiturates, the structure of which is that of barbituric acid substituted at N-1 by a methyl group and at C-5 by ethyl and phenyl groups.
- Manufacturing Process 46 parts metallic sodium was dissolved in 1000 parts absolute alcohol. The obtained solution was mixed with 264 parts of phenyl ethyl malonic acid diethyl ester and 80 parts of monomethyl urea and heated for 8 hours at reflux. Alcohol was distilled off, the residue was dissolved in water and neutralized with diluted sulfuric acid. N-Methylethylphenylbarbituric acid precipitated as a powder. It was filtered off, washed to neutral and dissolved in 50 parts of boiling alcohol. On cooling the obtained methylphenobarbital precipitated as the colorless prisms. MP: 176.5°C. This compound may be also prepared by condensation of equivalents of phenyl malonic ester and monomethyl urea, which was dissolved in above described solution of sodium ethylate.
- brand name Mebaral (Sterling Winthrop); Menta-Bal (Marion Merrell Dow).
- Therapeutic Function Anticonvulsant
- General Description Mephobarbital, 3-methyl-5-ethyl-5-phenylbarbituric acid (metharbital), is metabolicallyN-demethylated to phenobarbital, which many consider toaccount for almost all of the activity. Its principal use is asan anticonvulsant.
-
Clinical Use
Mephobarbital is a barbiturate-derivative AED with a pKa of 7.7 (log P = 1.84 at pH 7.4). Approximately 50% of an oral dose of
mephobarbital is absorbed from the gastrointestinal tract. The plasma concentrations required for its therapeutic effects are
unknown. The principal route of mephobarbital metabolism is N-demethylation by the liver to form phenobarbital, which may be
excreted in the urine unchanged and as its p-hydroxy metabolite and glucuronide or sulfate conjugates.
Conversion to the 4-hydroxy metabolite is stereoselective, being catalyzed by either CYP2C19 (R-enantiomer) or CYP2B6
(S-enantiomer); individuals who are CYP2C19 poor metabolizers show decreased clearance. Approximately 75% of a
single oral dose of mephobarbital is converted to phenobarbital. It has not been determined whether mephobarbital contributes
to the antiseizure effect or whether it results from its active metabolite, phenobarbital. Similarly, it is unclear whether
mephobarbital, like phenobarbital, is a potent inducer of the enzymes involved in the metabolism of other drugs, but because
the drug is chemically and pharmacologically similar to phenobarbital and is metabolized to phenobarbital, this possibility is
likely.
Mephobarbital is less commonly used in the treatment of generalized and partial seizures. Like phenobarbital, it is classified as a long-acting barbiturate. No evidence exists that it is more effective than phenobarbital in equivalent doses; however, it may be less sedating in children. - Safety Profile Poison by ingestion and intraperitoneal routes. A human teratogen by an unspecified route with developmental abnormalities of the cardovascular (circulatory) system. When heated to decomposition it emits toxic NOx. See also BARBITURATES.
MEPHOBARBITAL Preparation Products And Raw materials
Raw materials
Preparation Products
Global(0)Suppliers
115-38-8, MEPHOBARBITALRelated Search:
- Benzobarbital difebarbamate MEPHOBARBITAL Eterobarb febarbamate Barbituric acid Phenobarbital 5-PHENYL-PYRIMIDINE dimethylphenobarbital N-Methoxymethyl Phenobarbital PHENOBARBITAL-1-BUTYRIC ACID Mephobarbital-d3 (S)-Mephobarbital (R)-Mephobarbital Diphenylhydantoin and Mephobarbital(1:2) MEPHOBARBITAL CONTROLLED SUBSTANCE CIV USP(CRM STANDARD) 5-ETHYL-1-(2-FLUOROBENZOYL)-5-PHENYLPYRIMIDINE-2,4,6(1H,3H,5H)-TRIONE ADIPHENE
- Pharmaceuticals
- Intermediates & Fine Chemicals
- Heterocycles
- Aromatics
- METHYLPHENOBARBITAL (1.0MG/ML IN ACETONITRILE)
- METHYLPHENOBARBITAL[受控]
- 甲基苯巴比妥
- 甲苯巴比妥
- 5-乙基-1-甲基-5-苯基巴比妥酸
- 甲苯比妥
- 115-38-8
- Quetiapine Impurity 53
- Mephobarbital CIV (1386000)
- MEPHOBARBITAL USP/EP/BP
- Methylphenobarbital (1.0mg/ml in Acetonitrile)
- (5R)-5-ethyl-1-methyl-5-phenyl-1,3-diazinane-2,4,6-trione
- (RS)-Methylphenobarbital
- Mephobarbital CIV (250 mg)
- Methylphenobarbital (CIV), BP, EP
- 5-ethyl-1-methyl-5-phenyl-1,3-diazinane-2,4,6-trione
- Promitone
- MEPHOBARBITAL,BP,EP
- METHYLPHENOBARITAL
- Prominal
- Phenobarbital, mono-methyl
- phenmiton
- Phemitone
- Phemiton
- Phemetone
- N-Methylphenolbarbitol
- N-Methylphenobarbital
- n-methylethylphenylbarbituricacid
- N-Methylethylphenylbarbituric acid
- N-Methyl-5-phenyl-5-ethylbarbituric acid
- N-Methyl-5-phenyl-5-ethylbarbital
- n-ethylmethylphenylbarbituricacid
- N-Ethylmethylphenylbarbituric acid
- Morbusan
- Metyna
- Metylfenemal
- methylphenylbarbituricacid
- Methylphenylbarbituric acid
- methylphenolbarbital
- methylphenobarbitone
- Methyl-calminal
- Methyl phenobarbitone
- Mephytal
- Mephobsrbital
- Mephobarbitone
- Menta-bal
- Meberal
- Mebaral
- isonal(roussel)
- Isonal (Roussel)
- Hexahydropyrimidine-2,4,6-trione,1-methyl-5-ethyl-5-phenyl-
- Enphenemal
- Enfenemal
- barbituricacid,5-ethyl-1-methyl-5-phenyl-