Description
2’-
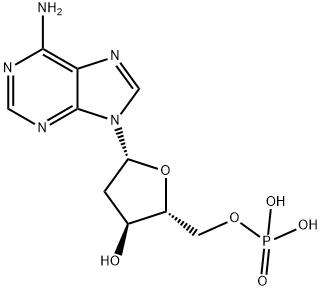
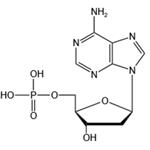
Deoxyadenosine-
5''-
monophosphate is a derivative of the nucleic acid, AMP, in which the hydroxyl group on the 2'' carbon of the pentose has been reduced to a hydrogen atom. It has been used in the synthesis of novel photoaffinity labels for incorporation into DNA and to study adenosine-
based interactions during DNA synthesis and DNA damage.
Definition
ChEBI: 2'-deoxyadenosine 5'-monophosphate is a purine 2'-deoxyribonucleoside 5'-monophosphate having adenine as the nucleobase. It has a role as a fundamental metabolite. It is a purine 2'-deoxyribonucleoside 5'-monophosphate and a 2'-deoxyadenosine 5'-phosphate. It is a conjugate acid of a 2'-deoxyadenosine 5'-monophosphate(2-).
References
[1] M ZOFALL B B. Two novel dATP analogs for DNA photoaffinity labeling.[J]. Nucleic Acids Research, 2000, 28 21: 4382-4390. DOI:
10.1093/nar/28.21.4382[2] De Souza, F.I., Zumiotti, A.V., and Da Silva, C.F. Neuregulins 1-α and 1-β on the regeneration the peripheral nerves[J]. Acta Ortop Bras.
[3] V DUARTE C J B J G Muller. Insertion of dGMP and dAMP during in vitro DNA synthesis opposite an oxidized form of 7,8-dihydro-8-oxoguanine.[J]. Nucleic Acids Research, 1999, 27 2: 496-502. DOI:
10.1093/nar/27.2.496