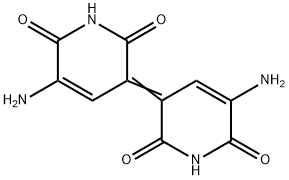
indigoidine
- Product Nameindigoidine
- CAS2435-59-8
- CBNumberCB92334234
- MFC10H8N4O4
- MW248.19
- MOL File2435-59-8.mol
Chemical Properties
| Boiling point | 285.6±40.0 °C(Predicted) |
| Density | 1.637±0.06 g/cm3(Predicted) |
| pka | 5.88±0.40(Predicted) |
indigoidine Chemical Properties,Usage,Production
Description
Indigoidine is a blue-violet organic pigment related to the Azaquinones group and synthesized by bacterial strains. It is biosynthesized by bacterial species such as Streptomyces chromofuscus,E. coli,and Corynebacterium insidiosum (Gangulyet al. 2019). It is used as a food colorant in cereal, baking, and ice-cream industries.Uses
Indigoidine natural blue dye is a promising alternative to the synthetic dyes used to colored jeans, leather, food, beverages, cosmetics and paper. The blue color of the indigoidine compound is bright and sustainable. As a natural dye, it has promising health benefits as an antioxidant and antimicrobial actor. Because of a new purification process patented with the compound, indigoidine natural blue dye is safe to use in food and drinks.Definition
ChEBI: Indigoidine is a member of the class of pyridone that is a dimeric blue pigment biosynthesised from L-glutamine. It has a role as a bacterial metabolite and a biological pigment. It is a pyridone, a ring assembly, a dicarboximide and an enamine. It derives from a L-glutamine.Definition
Pigment characterization:Identification of interrupted genes in the nonpigmented mutants suggested that the blue compound produced by Y4I was indigoidine, a bicyclic 3,3′-bipyridyl molecule synthesized by the cyclization of two glutamine molecules. The absorbance spectrum of the purified compound in DMSO has a peak at 612 nm, which has been previously reported as the absorbance maximum of indigoidine. Profiles of the Y4I variant show that absorbance at this wavelength for the clpA::Tn5 mutant is approximately five times higher than that of the wild-type strain[1].
Biosynthesis
Indigoidine is a water-insoluble blue pigment that was first isolated from phytopathogenic Erwinia as a powerful radical scavenger that enables phytopathogens to tolerate oxidative stress, organic peroxides, and superoxides during the plant defense response due to its structure of carbon–carbon double bonds conjugated with a carbonyl group. This bacterial pigment shows a bright blue color similar to that of indigo. Several different strains are reported to produce it. Indigoidine is assembled from two units of L-glutamine by a nonribosomal peptide synthetase (e.g. IndC from Erwinia chrysanthemi and Streptomyces aureofaciens CCM 3239, BpsA from Streptomyces lavendulae and Sc-indC from Streptomyces chromofuscus ATCC 49982) (Figure 1.7b). Recently, an indigoidine biosynthetic gene cluster was located in the genome of S. chromofuscus ATCC 49982. The gene cluster is silent and consists of five open reading frames, called orf1, Sc-indC, Sc-indA, Sc-indB, and orf2. Sc-IndC was functionally characterized as an indigoidine synthase through heterologous expression of the enzyme in both Streptomyces coelicolor CH999 and E. coli BAP1. The titer of indigoidine in E. coli BAP1 was reported to be 2.78 g/L under optimized conditions. Its production was dramatically increased (by 41.4%/3.93 g/L) when Sc-IndB was co-expressed with it in E. coli BAP1 (Yu et al. 2013). In order to further improve production, a glutamine synthetase gene was amplified from E. coli and co-expressed with Sc-indC and Sc-indB in E. coli BAP1. At 2.5 mM (NH4)2HPO4, the titer can reach 7.08±0.11 g/L (Xu et al. 2015). This provides a green, efficient production process for this promising blue dye.Biological Activity
Research has suggested that indigoidine protects D. dadantii by neutralizing reactive oxygen species generated by the plant during bacterial infection. This was the only reported biological role for indigoidine [1].Solubility in water
Indigoidine is found in two redox states, an oxidized blue form which is insoluble in water and a reduced colorless form which is water soluble and historically referred to as leucoindigoidine[1].References
[1] W Nathan Cude. “Production of the antimicrobial secondary metabolite indigoidine contributes to competitive surface colonization by the marine roseobacter Phaeobacter sp. strain Y4I.” Applied and Environmental Microbiology 78 14 (2012): 4771–80.Preparation Products And Raw materials
indigoidine Suppliers
Global(13)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| +86-023-62871537 | sales@rosewachem.com | China | 2827 | 58 | |
| 18507340638 | 2335663646@qq.com | China | 197 | 58 | |
| 027-59210159 15377098680 |
1148280011@qq.com | China | 9950 | 58 | |
| 027-59768661 13297943853 |
1704244976@qq.com | China | 5005 | 58 | |
| 18551497631 18551497631 |
507167383@qq.com | China | 5026 | 58 | |
| 0513-66017706 18551496367 |
2186883463@qq.com | China | 4773 | 58 | |
| 027-59106191 13986034860 |
1400828855@qq.com | China | 8944 | 58 | |
| 18086118076; 18086118076 |
1784708432@qq.com | China | 4446 | 58 | |
| 18969655221 | 530532315@qq.com | China | 206 | 58 | |
| 0513-0513-66814573 16651336298 |
16651336298@163.com | China | 4227 | 58 |
2435-59-8, indigoidineRelated Search
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine