Synthesis
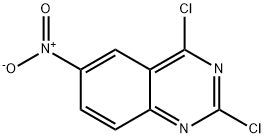
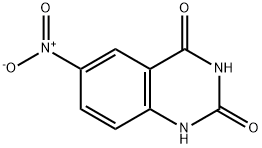
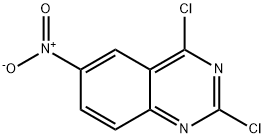
General procedure for the synthesis of 2,4-dichloro-6-nitroquinazoline from 2,4-dihydroxy-6-nitroquinazoline: To a solution of 6-nitroquinazoline-2,4(1H,3H)-dione (34 g, 164.14 mmol) in trichlorophosphorus (POCl3, 150 mL) was added N,N-dimethylaniline (60 g, 492.42 mmol). The reaction mixture was stirred at 110 °C for 5 hours. After completion of the reaction, the mixture was concentrated under reduced pressure and the remaining residue was neutralized with aqueous sodium bicarbonate. The aqueous phase was extracted with ethyl acetate (EtOAc, 80 mL x 3). The organic phases were combined, concentrated to dryness and the residue was purified by silica gel column chromatography (eluent: dichloromethane/petroleum ether = 2:1) to afford 2,4-dichloro-6-nitroquinazoline (5 g, yield: 13%) as a yellow solid.
References
[1] Patent: US2010/204226, 2010, A1. Location in patent: Page/Page column 61
[2] Bioorganic and Medicinal Chemistry, 2011, vol. 19, # 9, p. 2797 - 2807
[3] Patent: CN102250075, 2016, B. Location in patent: Paragraph 0395; 0414; 0415
[4] Patent: WO2018/114700, 2018, A1. Location in patent: Page/Page column 22; 53
[5] Patent: WO2015/200534, A2. Location in patent: Paragraph 00414