Synthesis
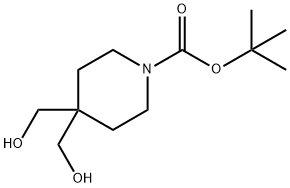
The general procedure for the synthesis of tert-butyl 2-oxa-7-azaspiro[3.5]nonane-7-carboxylate from tert-butyl 4,4-bis(hydroxymethyl)piperidine-1-carboxylate was as follows: to a solution of tert-butyl 4,4-bis(hydroxymethyl)piperidine-1-carboxylate (60.0 g, 244.58 mmol) in anhydrous THF (1.2 L) was slowly added n-butyllithium ( nBuLi, 97.8 mL, 0.245 mol, 2.5 M) and the reaction mixture was stirred for 0.5 h at 0 °C. Subsequently, a solution of anhydrous THF (600 mL) of p-toluenesulfonyl chloride (TsCl, 46.6 g, 0.245 mmol) was added drop-wise, and the reaction process was monitored by thin-layer chromatography (TLC) until the feedstock was completely consumed, and stirring was continued for 1.5 hr at 0 °C. Again n-butyllithium (nBuLi, 97.8 mL, 0.245 mmol, 2.5 M) was added to the reaction mixture at 0 °C and stirred for 0.5 hours at 0 °C. Subsequently, the reaction mixture was warmed to 60 °C and stirred for 1 hour. Upon completion of the reaction, the reaction was quenched by addition of saturated ammonium chloride (NH4Cl, 400 mL) solution at 0 °C. After separation of the organic layer, the aqueous layer was extracted with ethyl acetate (EtOAc, 300 mL x 3). The combined organic phases were washed with brine (800 mL × 2), dried over anhydrous sodium sulfate (Na2SO4) and concentrated under reduced pressure to give the crude product (65 g). The crude product was purified by fast column chromatography (eluent: petroleum ether (PE):ethyl acetate (EtOAc) = 30:1 to 10:1) to afford the target compound 2-oxa-7-azaspiro[3.5]nonane-7-carboxylic acid tert-butyl ester (37.0 g, 66% yield) as a white solid. The structure of the product was confirmed by 1H NMR (CDCl3) and mass spectrometry (MS): 1H NMR (CDCl3): δ1.47 (s, 9H), 1.83 (t, 4H, J=5.6Hz), 3.35 (t, 4H, J=5.6Hz), 4.46 (s, 4H); MS: 127.9 [M-Boc+H]+.
References
[1] Tetrahedron Letters, 2011, vol. 52, # 26, p. 3266 - 3270
[2] Patent: CN104557871, 2017, B
![2-Oxa-7-azaspiro[3.5]nonane-7-carboxylic acid, 1,1-dimethylethyl ester Structure](https://www.chemicalbook.com/CAS/GIF/240401-27-8.gif)

![2-Oxa-7-azaspiro[3.5]nonane-7-carboxylic acid, 1,1-dimethylethyl ester](/CAS/GIF/240401-27-8.gif)
