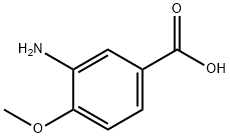
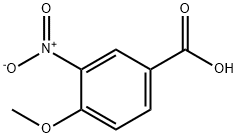
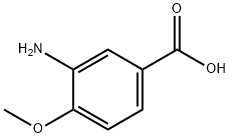
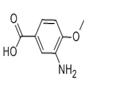
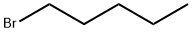Uses
It is an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuffs.
Synthesis
The general procedure for the synthesis of 3-amino-4-methoxybenzoic acid from 1-bromopentane and 4-methoxy-3-nitrobenzoic acid was as follows: 4-methoxy-3-nitrobenzoic acid (5 g, 25.4 mmol), DMF (30 ml), potassium carbonate (5.53 g, 40 mmol) and 1-bromopentane (4 ml, 32.3 mmol) were mixed at 100 °C and The reaction was stirred for 1.5 hours. After completion of the reaction, the reaction mixture was filtered to remove the inorganic salts, followed by evaporation of DMF under reduced pressure. ethyl acetate (100 ml) was added to the residue, the mixture was washed three times with saturated brine (30 ml) and dried with anhydrous magnesium sulfate. The desiccant was removed by filtration and the filtrate was concentrated under reduced pressure. To the resulting residue was added ethanol (150 ml) and 10% palladium-carbon catalyst (0.5 g) and stirred in a stream of hydrogen for 5.5 hours at room temperature. At the end of the reaction, the palladium-carbon catalyst was removed by filtration and the filtrate was concentrated under reduced pressure. Finally, the residue was purified by column chromatography (eluent: hexane/ethyl acetate = 3/1) to afford 3-amino-4-methoxybenzoic acid (5.98 g, 99% yield, two-step reaction) as colorless crystals. The product was confirmed by 1H-NMR (CDCl3) and FABMS characterization.