Synthesis
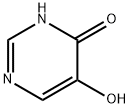
Example 9: Synthesis of 5-hydroxypyrimidin-4(3H)-one
To a mixed solution containing water (76 cm3) and concentrated ammonia (7.6 cm3) was added Compound 5c (11.0 g) followed by Ruanne nickel (40.0 g). The reaction mixture was heated to reflux for 4 hours. After completion of the reaction, the mixture was cooled to room temperature and filtered. All volatile solvents were removed using a rotary evaporator and the resulting residue was redissolved in methanol. Addition of ether to this methanol solution induced precipitation of the target product in the form of pink crystals. The crystals were collected and dried under vacuum at 100 °C to afford 5-hydroxypyrimidin-4(3H)-one (2.5 g, 46% yield).
Product Characterization:
- Melting point: 265-267 °C
- 1H NMR (300 MHz, DMSO-d6) δ: 7.40 (1H, s), 7.67 (1H, s)
- IR (cm1): 1640, 1600, 1360, 1300, 1270, 1100, 930, 880, 790, 780, 615
- MS (EI): m/z 112 (100%, C4H4N2O2, M)
References
[1] Tetrahedron, 2006, vol. 62, # 23, p. 5469 - 5473
[2] Patent: WO2007/116412, 2007, A1. Location in patent: Page/Page column 11; 14
![4(1H)-Pyrimidinone, 2,3-dihydro-5-[(tetrahydro-2H-pyran-2-yl)oxy]-2-thioxo-](/CAS/20210305/GIF/894421-78-4.gif)