Synthesis
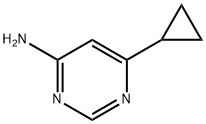
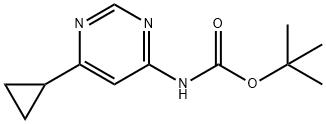
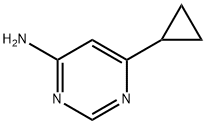
Step 2: A mixture of tert-butyl 6-cyclopropylpyrimidin-4-ylcarbamate (256 mg, 0.764 mmol) and tert-butyl 6-cyclopropylpyrimidin-4-ylcarbamate (10 mg, 0.043 mmol) was dissolved in a dioxane solution of 4N HCl (3.0 mL, 12 mmol) and heated for 3 hr at 55 °C. Subsequently, additional dioxane solution of 4N HCl (1.0 mL, 4 mmol) was added and the reaction mixture was warmed up to 70°C to continue heating for 1.5 hours, followed by stirring at room temperature overnight. Upon completion of the reaction, the mixture was partitioned between saturated aqueous sodium bicarbonate solution and ethyl acetate. The organic layer was washed with brine, dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure. The residue was purified by rapid chromatography on silica gel (eluent: dichloromethane solution of 2-5% methanol) to give 6-cyclopropylpyrimidin-4-amine (84 mg, 78% yield). The product was characterized by NMR (400 MHz, methanol-d4): δ 8.17 (d, J = 1.1 Hz, 1H), 6.36 (d, J = 1.2 Hz, 1H), 4.61 (br s, 2H), 1.90-1.81 (m, 1H), 1.03-0.94 (m, 4H).