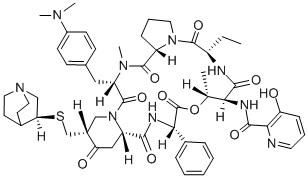
Description
Quinupristin is a streptogramin antibiotic that blocks peptide bond formation and peptide extension in bacteria. Quinupristin is usually combined with another streptogramin antibiotic, dalfopristin , to produce quinupristin-dalfopristin complex , known commercially as Synercid. While both quinupristin and dalfopristin have bacteriostatic effects, they act synergistically to kill Gram-positive bacteria, as well as some Gram-negative and anaerobic bacteria.
Chemical Properties
White to off-white solid, soluble in ethanol, methanol, DMSO and water, unstable in aqueous solution.
Uses
Semisynthetic depsipeptide type I streptogramin. An antibacterial agent.
Uses
Quinupristin is a semi-synthetic analogue of virginiamycin B (ostreogrycin B, pristinamycin IA, streptogramin B) formed by a Mannich condensation and elimination to generate an exocyclic methylene alpha to the ketone of the 4-piperidinone. Addition of quinucidinylthiol to the methylene group affords quinupristin. The structural changes provide a more hydrophobic compound with a readily ionisable group for generating a salt. Quinupristin is used commercially in synergistic combination with dalfopristin (30:70). There is little published data on the synthesis, biological or antibiotic activity of quinupristin alone, however the combination product is highly effective, including activity against antibiotic resistant strains.
Uses
Quinupristin is a semi-synthetic analogue of virginiamycin B (ostreogyrcin B, pristinamycin IA, streptogramin B) formed by a Mannich condensation and elimination to generate an exocyclic methylene alpha to the ketone of the 4-piperidinone. Addition of quinucidinylthiol to the methylene group affords quinupristin. The structural changes provide a more hydrophobic compound with a readily ionisable group for generating a salt. Quinupristin is used commercially in synergistic combination with dalfopristin (30:70). There is little published data on the synthesis, biological or antibiotic activity of quinupristin alone, however the combination product is highly effective, including activity against antibiotic resistant strains.
References
[1] MICHAEL A. KOHANSKI James J C Daniel J Dwyer. How antibiotics kill bacteria: from targets to networks[J]. Nature Reviews Microbiology, 2010, 8 6: 423-435. DOI:
10.1038/nrmicro2333[2] JONAS NOESKE. Synergy of streptogramin antibiotics occurs independently of their effects on translation.[J]. Antimicrobial Agents and Chemotherapy, 2014: 5269-5279. DOI:
10.1128/aac.03389-14[3] WILSON D N. The A-Z of bacterial translation inhibitors.[J]. Critical Reviews in Biochemistry and Molecular Biology, 2009, 44 6: 393-433. DOI:
10.3109/10409230903307311