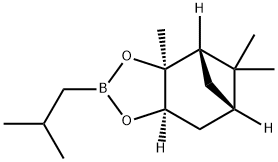
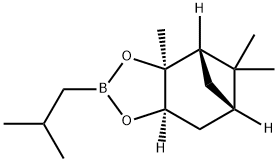
Synthesis
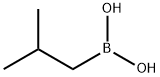
GENERAL STEPS: A reaction mixture of (1S,2S,3R,5S)-(+)-2,3-pinanediol (6.9 g) with isobutylboronic acid (4.4 g) in diethyl ether (49.3 mL) was stirred at room temperature for 23 hours. After the reaction was completed, anhydrous sodium sulfate (16.5 g) was added to dry the reaction mixture. The sodium sulfate was removed by filtration and the filter cake was washed with diethyl ether (69.4 mL). The organic phases were combined and concentrated under reduced pressure to give a colorless oily product (9.9 g, 100% GC purity, 97.2% yield). Mass spectrum (MS, m/z): 236, 221, 195, 167, 140, 134, 83, 67, 55, 43; 1H-NMR (DMSO-d6, δ): 4.27 (1H, dd, J = 2.1 Hz, 8.7 Hz), 2.30 (1H, m), 2.18 (1H, m), 1.96 (1H, t, J = 5.4 Hz ), 1.86 (1H, m), 1.79 (1H, sx, J = 6.8 Hz), 1.69 (1H, m), 1.30 (3H, s), 1.25 (3H, s), 1.02 (1H, d, J = 10.6 Hz), 0.9 (3H, d, J = 6.6 Hz), 0.81 (3H, s), 0.69 (2H, m).
References
[1] Patent: WO2009/4350, 2009, A1. Location in patent: Page/Page column 9
[2] Bioorganic and Medicinal Chemistry, 2013, vol. 21, # 4, p. 1018 - 1029

![Bicyclo[3.1.1]heptane-2,3-diol, 6,6-dimethyl-, (1α,2β,3α,5α)- (9CI)](/CAS/20210305/GIF/57022-61-4.gif)