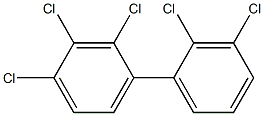
AROCLOR 1254, 1X1ML, ISO, 1000UG/ML
- Product NameAROCLOR 1254, 1X1ML, ISO, 1000UG/ML
- CAS11097-69-1
- CBNumberCB6498490
- MFC12H5Cl5
- MW326.4331
- MOL File11097-69-1.mol
- MSDS FileSDS
Chemical Properties
| Melting point | 95.86°C (estimate) |
| Boiling point | 412.89°C (rough estimate) |
| Density | 1.5000 (estimate) |
| vapor pressure | 3.22 at 25 °C (estimated using GC retention data, Foreman and Bidleman, 1985) |
| refractive index | 1.6200 (estimate) |
| Flash point | -12 °C |
| storage temp. | room temp |
| solubility | (wt %): Pyridine (114 at 31 °C), diglycol monoethyl ether (173 at 26 °C), methanol (15 at 26 °C), ethanol (10 at 27 °C) (Monsanto, 1960) |
| Water Solubility | 43ug/L(20 ºC) |
| Henry's Law Constant | 83.7 at 25 °C (gas stripping-UV spectrophotometry, Warner et al., 1987) |
| Exposure limits | Potential occupational carcinogen. NIOSH REL: TWA 1 μg/m3, IDLH 5 mg/m3; OSHA PEL: TWA 0.5 mg/m3; ACGIH TLV: TWA 1 mg/m3 (adopted). |
| Stability | Stable. Highly flammable. Incompatible with strong oxidizing agents. Attacks some forms of plastics and rubber. |
| Major Application | environmental |
| InChI | 1S/C12H5Cl5/c13-8-3-1-2-6(10(8)15)7-4-5-9(14)12(17)11(7)16/h1-5H |
| InChIKey | AUGNBQPSMWGAJE-UHFFFAOYSA-N |
| SMILES | Clc1c(c(ccc1Cl)c2c(c(ccc2)Cl)Cl)Cl |
| EPA Substance Registry System | Aroclor 1254 (11097-69-1) |
| UNSPSC Code | 12000000 |
Safety
| Symbol(GHS) |
  
|
|||||||||
| Signal word | Warning | |||||||||
| Hazard statements | H302-H373-H410 | |||||||||
| Precautionary statements | P273-P301+P312+P330-P314 | |||||||||
| Hazard Codes | F,Xn,N,T | |||||||||
| Risk Statements | 11-38-50/53-65-67-33-62-51/53-48/20-36/37/38-22-39/23/24/25-23/24/25-40-37/38-20/21/22-45 | |||||||||
| Safety Statements | 9-16-29-33-60-61-62-35-36/37-36-26-45-7-23-53 | |||||||||
| RIDADR | 2315 | |||||||||
| OEB | E | |||||||||
| OEL | TWA: 0.001 mg/m3 [*Note: The REL also applies to other PCBs.] | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | TQ1360000 | |||||||||
| HazardClass | 9 | |||||||||
| PackingGroup | II | |||||||||
| Storage Class | 6.1C - Combustible, acute toxic Cat.3 toxic compounds or compounds which causing chronic effects | |||||||||
| Hazard Classifications | Aquatic Chronic 2 Carc. 1B STOT RE 2 | |||||||||
| Hazardous Substances Data | 11097-69-1(Hazardous Substances Data) | |||||||||
| Toxicity | Acute oral LD50 for rats 1,010 mg/kg (quoted, RTECS, 1985). | |||||||||
| IDLA | 5 mg/m3 | |||||||||
| NFPA 704: |
|