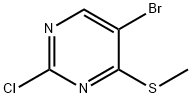
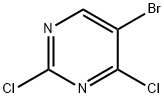
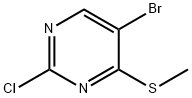
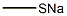Synthesis
General procedure for the preparation of 5-bromo-2-chloro-4-(methylthio)pyrimidine: 2 g of sodium methanethiol (28.5 mmol, 1 eq.) and 6.5 g of 5-bromo-2,4-dichloropyrimidine (28.5 mmol, 1 eq.) were reacted at room temperature with stirring for 24 hr in 50 mL of anhydrous acetonitrile. Upon completion of the reaction, the mixture was poured into water and extracted with dichloromethane, the organic phase was dried over anhydrous sodium sulfate and subsequently concentrated under reduced pressure to remove the solvent. The crude product was purified by recrystallization from hexane to give 4 g of the target compound (70% yield).1H-NMR (400 MHz, CDCl3) data: δ 8.31 (s, 1H), 2.59 (s, 3H).
References
[1] Patent: EP1878726, 2008, A1. Location in patent: Page/Page column 36
[2] Patent: US2008/176866, 2008, A1. Location in patent: Page/Page column 8
[3] Patent: WO2007/71455, 2007, A1. Location in patent: Page/Page column 252-253
[4] Patent: US2007/232632, 2007, A1. Location in patent: Page/Page column 109
[5] Bioorganic and Medicinal Chemistry Letters, 2007, vol. 17, # 22, p. 6206 - 6211