Synthesis
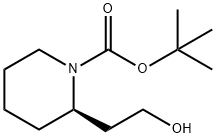
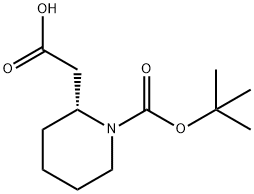
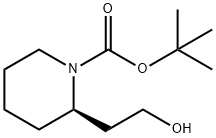
General procedure for the synthesis of (R)-1-N-Boc-piperidin-2-yl-ethanol from (R)-2-(1-(tert-butoxycarbonyl)piperidin-2-yl)acetic acid: to a solution of (R)-2-(1-(tert-butoxycarbonyl)piperidin-2-yl)acetic acid (2.0 g, 8.7 mmol) in anhydrous CH2Cl2 (20 mL) was added drop-wise a lithium aluminium hydride 2.0 M THF solution (7.23 mL, 13.05 mmol). The reaction mixture was heated to 55 °C under argon protection and stirred for 4 hours. Upon completion of the reaction, the mixture was cooled to room temperature, diluted with ethyl acetate (20 mL) and the reaction was quenched with saturated NaHCO3 solution (3 mL). Subsequently, the solution was concentrated under reduced pressure. CH2Cl2 (20 mL) was added to the residue, and the organic layer was washed sequentially with water and brine, dried over anhydrous Na2SO4, and concentrated under reduced pressure to afford (R)-1-N-Boc-piperidin-2-ethanol (1.72 g, 8.0 mmol) as a light-colored oil in 91.9% yield. The same method was applied to synthesize intermediates 7-10.
References
[1] European Journal of Medicinal Chemistry, 2018, vol. 145, p. 790 - 804