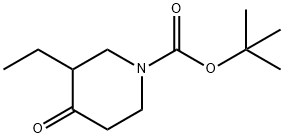
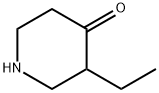
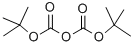
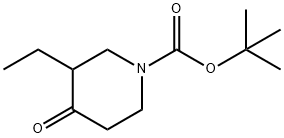
Synthesis
The general procedure for the synthesis of tert-butyl 3-ethyl-4-oxopiperidine-1-carboxylate from 3-ethylpiperidin-4-one and di-tert-butyl dicarbonate was carried out as follows: the reaction was carried out by suspending 3-ethylpiperidin-4-one (2.90 g, 10.2 mmol) in 6N hydrochloric acid (85 ml) at reflux for 16 hours. Upon completion of the reaction, the reaction solution was concentrated and residual water was removed by azeotroping three times with isopropanol to give a yellow solid. The solid was dissolved in a 1:1 solvent mixture of dichloromethane and methanol (50 ml) and triethylamine (3.08 g, 4.3 ml, 30.5 mmol) and di-tert-butyl dicarbonate (3.32 g, 15.2 mmol) were added sequentially. The reaction mixture was stirred at 23 °C for 16 h. The reaction solution was subsequently concentrated. To the concentrate was added 0.5N sodium hydroxide solution (50 ml) and extracted with dichloromethane. The organic phases were combined, dried with anhydrous magnesium sulfate, filtered and concentrated. Finally, purification by silica gel column chromatography (eluent: 10% ethyl acetate-hexane) gave 1.92 g (8.46 mmol, 83% yield) of the target product tert-butyl 3-ethyl-4-oxopiperidine-1-carboxylate as a colorless oil. Mass spectrum (ESI+): m/z 228 [M+1]+.