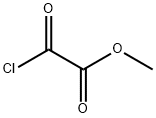
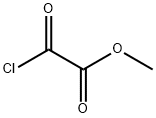
Uses
Bicyclo[1.1.1]pentane-1,3-dicarboxylic Acid, 1-methyl Ester acts as a reagent for the synthesis and in the biopharmaceutical evaluation of imatinib analogs. Also functions as a reagent for the preparation and in biological evaluation of novel bicyclo[1.1.1]pentane-based acidic amino acids as glutamate receptors ligands It is a COVID19-related research product.
Synthesis
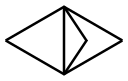
The general procedure for the synthesis of methyl bicyclo[1.1.1]pentane-1-carboxylate-3-carboxylate from oxalyl chloride monomethyl ester and the compound (CAS:35634-10-7) was as follows: first, a solution of [1.1.1]propeller in Et2O (500 mL; 161 mmol, 1 eq.) was prepared independently, along with ethyl chloroacetate (16.3 mL, 177 mmol, 1.1 eq.) solution in Et2O (49.7 mL). The entire reactor was flushed for 5 min using pure Et2O (pump 1: 4.0 mL/min, pump 2: 0.5 mL/min). Subsequently, a solution of reagent (2) was injected at a flow rate of 4.0 mL/min at 0 to -80 °C, followed by a solution of reagent (3) at a flow rate of 0.5 mL/min at room temperature. The photoreactor was placed in an ice bath (volume 58 mL; FEP tube rotated over a lamp). After the reaction mixture left the photoreactor, it was quenched using an alkaline aqueous solution of NaOH, KOH, NaHCO3, or KHCO3. After vigorous stirring for 2 h, the aqueous layer was separated and acidified to pH = 1. Subsequently, the mixture was extracted with DCM (3 × 500 mL). The combined organic layers were dried over MgSO4, filtered and concentrated under reduced pressure to afford 3-(methoxycarbonyl)bicyclo[1.1.1]pentane-1-carboxylic acid (9, 17.5 g, 103 mmol, 63% yield), the product being a pale yellow solid.
![Bicyclo[1.1.1]pentane-1,3-dicarboxylic acid, MonoMethyl ester Structure](https://www.chemicalbook.com/CAS/GIF/83249-10-9.gif)

![Tricyclo[1.1.1.01,3]pentane](/CAS/GIF/35634-10-7.gif)
![Bicyclo[1.1.1]pentane-1,3-dicarboxylic acid, MonoMethyl ester](/CAS/GIF/83249-10-9.gif)