Synthesis
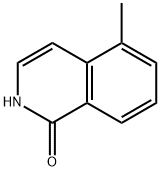
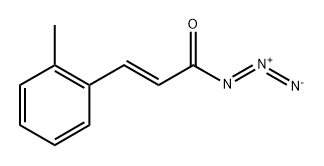
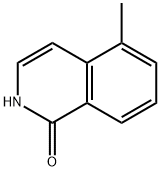
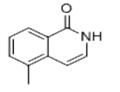
Synthesis of Intermediate-3: 5-Methylisoquinolin-1(2H)-one: o-Methylcinnamoyl azide (Intermediate-2) (10 g, 53.5 mmol) was dissolved in toluene (100 mL) with stirring and heated to 110±2°C for 1 hour. After completion of the reaction, the toluene solvent was removed under reduced pressure, 1,2-dichlorobenzene (DCB) (100 mL) and iodine (catalytic amount) were added, and the reaction mixture was heated to 180±2°C and kept reacting overnight. At the end of the reaction, the solvent was removed under reduced pressure and the product was purified by recrystallization (ethyl acetate: hexane = 90:10) to afford the target compound 5-methylisoquinolin-1(2H)-one (4 g, 45% yield).
References
[1] Patent: US2013/178457, 2013, A1. Location in patent: Paragraph 0175
[2] Patent: WO2013/5168, 2013, A2. Location in patent: Page/Page column 33
[3] Journal of the Chemical Society - Perkin Transactions 1, 1997, # 8, p. 1147 - 1156
[4] Journal of Heterocyclic Chemistry, 2001, vol. 38, # 4, p. 961 - 964