Synthesis
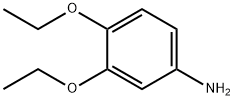
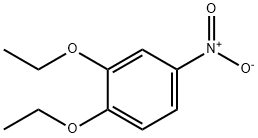
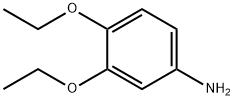
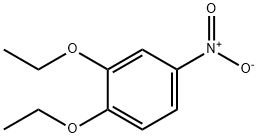
The general procedure for the synthesis of 3,4-diethoxyaniline from 3,4-diethoxynitrobenzene was as follows: 3,4-diethoxynitrobenzene (6.30 g, 29.9 mmol), 10% palladium-carbon catalyst (600 mg), and anhydrous ethanol (300 mL) were added to a reaction flask, and the reaction was stirred for 6 hours under hydrogen atmosphere. Upon completion of the reaction, the insoluble palladium-carbon catalyst was removed by filtration and the filtrate was concentrated under reduced pressure to afford 3,4-diethoxyaniline as an oily product in a yield of 5.02 g (92.9% yield). The product was characterized by 1H-NMR (CDCl3) with chemical shifts δ: 1.34 to 1.46 (6H, m), 3.94 to 4.08 (4H, m), 6.21 (1H, d, J = 2.8,8.4 Hz), 6.30 (1H, d, J = 2.6 Hz), 6.73 (1H, d, J = 8.4 Hz).
References
[1] Patent: EP1437344, 2004, A1. Location in patent: Page 37
[2] Yuki Gosei Kagaku Kyokaishi, 1959, vol. 17, p. 166,169
[3] Chem.Abstr., 1959, p. 11280