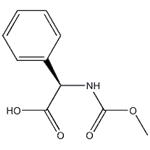
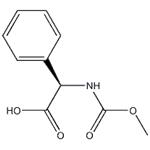
Synthesis
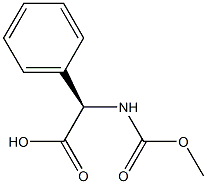
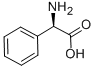
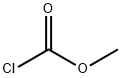
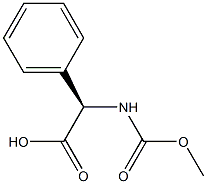
D-Phenylglycine (10.0 g, 66.1 mmol) and sodium hydroxide (21.2 g, 265 mmol) were dissolved in water (60 mL) at 0°C and stirred until completely dissolved. Subsequently, methyl chloroformate (10.2 mL, 133 mmol) was added slowly and dropwise over 20 minutes. The reaction mixture was continued to be stirred at 0°C for 1 hour. Upon completion of the reaction, it was acidified with concentrated hydrochloric acid (25 mL, 300 mmol) to pH < 2. The acidic aqueous phase was extracted with ethyl acetate (3 x 100 mL), the organic phases were combined and dried over anhydrous magnesium sulfate. After filtration to remove the desiccant, the organic phase was concentrated under reduced pressure to afford N-methoxycarbonyl-D-phenylglycine (Int-2a, 12.6 g, 91% yield), which could be used for subsequent reactions without further purification.