Description
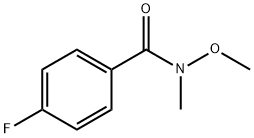
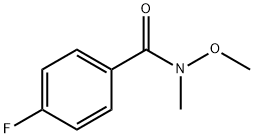
4-Fluoro-N-methoxy-N-methylbenzamide is a fluorinated aromatic. As an organofluorine compound, it can synthesise pyridyl heterocycles and photochromic compounds. 4-Fluoro-N-methoxy-N-methylbenzamide can form exocyclic fluorinating agents by reacting with iodides. It can form complexes with copper or zinc as a ligand and convert metal hydroxides into metal carbonyls as a carbonylative agent. As a phosphine, it can be converted into carboxylic acids through hydrolysis or oxidation.
Synthesis
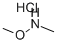
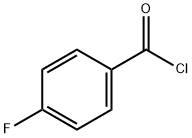
The general procedure for the synthesis of 4-fluoro-N-methoxy-N-methylbenzamide from dimethylhydroxylamine hydrochloride and 4-fluorobenzoyl chloride was as follows: 4-fluorobenzoyl chloride (15 g, 94.6 mmol) and N,O-dimethylhydroxylamine hydrochloride (10.1 g, 104 mmol) were dissolved in chloroform (200 mL) and stirred at room temperature. The reaction solution was cooled to 0 °C and pyridine (17.3 mL, 230 mmol) was added slowly. The reaction mixture was gradually warmed to room temperature and stirring was continued for 1 hour. After the reaction was completed, the mixture was poured into saturated sodium chloride solution (300 mL). The organic layer was separated and the aqueous layer was extracted with dichloromethane (3 x 100 mL). The organic layers were combined, washed with water (3 x 50 mL), dried over anhydrous sodium sulfate and subsequently concentrated under reduced pressure. The crude product was purified by vacuum distillation to afford the colorless liquid 4-fluoro-N-methoxy-N-methylbenzamide in 96% yield with a boiling point of 120 °C (0.3 mmHg). The characteristic absorption peaks of its infrared spectrum were 583, 905, 918, 1262, 1375, 1508, 1582, 1630, 2972, 3274 cm^-1; the NMR hydrogen spectral data were δH 3.34 (3H, s, CH3), 3.52 (3H, s, OCH3), 7.08 (2H, m, Ar-H), and 7.73 (2H, m , Ar-H).
References
[1] Bioorganic and Medicinal Chemistry, 1997, vol. 5, # 1, p. 49 - 64
[2] Bioorganic and Medicinal Chemistry Letters, 2014, vol. 24, # 5, p. 1352 - 1357
[3] Organic and Biomolecular Chemistry, 2013, vol. 11, # 20, p. 3337 - 3340
[4] Journal of Organic Chemistry, 2007, vol. 72, # 15, p. 5828 - 5831
[5] Patent: US5616596, 1997, A