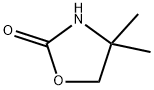
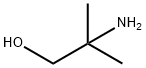
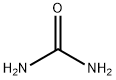
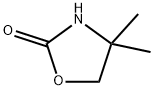
Synthesis
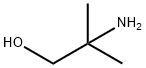
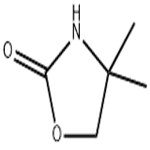
4,4-Dimethyloxazolidin-2-one (DMO) was synthesized as follows: 2-amino-2-methyl-1-propanol (10.0 g, 112 mmol) was mixed with urea (13.5 g, 224 mmol), and the mixture was heated and reacted for 1 hour at 170-180 °C, followed by heating up to 210-220 °C and continued heating for 1 hour. After completion of the reaction, it was cooled to room temperature and the reaction mixture was dissolved in water (50 mL). Dichloromethane (DCM, 5 x 50 mL) extraction was performed with the aqueous solution. The organic phases were combined, dried with anhydrous sodium sulfate (Na2SO4), filtered and concentrated under reduced pressure to afford 4,4-dimethyloxazolidin-2-one 10.3 g in 80% yield. The product was identified by 1H NMR (CDCl3): δ 6.13 (br s, 1H, NH), 4.07 (s, 2H, CH2), 1.32 (s, 6H, 2 CH3) ppm; 13C NMR (CDCl3) δ 159.4,77.1,55.4,27.7 ppm.
References
[1] Asian Journal of Chemistry, 2011, vol. 23, # 2, p. 929 - 930
[2] Patent: WO2015/68159, 2015, A2. Location in patent: Paragraph 00121-00122