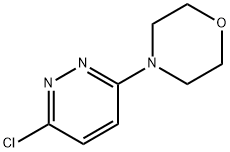
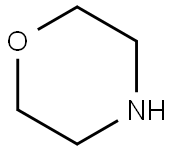
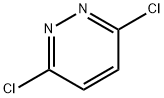
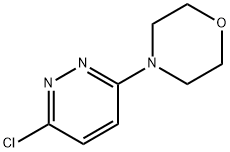
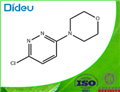
Synthesis
GENERAL METHOD: Anhydrous ethanol (5 mL) and 3,6-dichloropyridazine (3.36 mmol) were added to a 50 mL round-bottomed flask, followed by triethylamine (5.03 mmol) and morpholine (5.03 mmol). The reaction mixture was refluxed in ethanol (for 3,6-dichloropyridazine and 2,6-dichloropyrazine) or stirred at room temperature (for 2,4-dichloropyrimidine). The reaction process is monitored by gas chromatography (GC). After the starting material 3,6-dichloropyridazine was completely consumed, the reaction mixture was poured into saturated ammonium chloride solution (20 mL) and extracted with dichloromethane (3 x 20 mL). The organic layers were combined, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude product was ground with petroleum ether and filtered through a Büchner funnel to give the pure 4-(6-chloropyridazin-3-yl)morpholine.
References
[1] Tetrahedron, 2015, vol. 71, # 29, p. 4859 - 4867
[2] Journal of Organic Chemistry, 2013, vol. 78, # 2, p. 370 - 379
[3] European Journal of Medicinal Chemistry, 2015, vol. 95, p. 277 - 301
[4] Patent: WO2005/92899, 2005, A1. Location in patent: Page/Page column 87
[5] Bioorganic and Medicinal Chemistry, 2007, vol. 15, # 4, p. 1586 - 1605