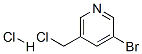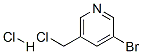Synthesis
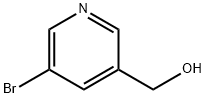
The general procedure for the synthesis of 3-bromo-5-(chloromethyl)pyridine hydrochloride from 3-bromopyridine-5-methanol was as follows: (5-bromopyridin-3-yl)methanol (XLV) (1.54 g, 8.2 mmol) was dissolved in a dioxane solution (10 mL) of 4 M HCl at 0 °C, followed by evaporation treatment. The resulting residue was dissolved in SOCl2 (4 mL) and reacted under reflux conditions for 2 hours. Upon completion of the reaction, SOCl2 was removed and the residue was ground with hexane to afford the HCl salt of 3-bromo-5-(chloromethyl)pyridine (XLVT) as a brown solid (1.30 g, 5.4 mmol, 66% yield). The product was used for subsequent experiments without further purification.ESIMS analysis showed that the product C6H5BrClN had a m/z of 206 (M + H).
References
[1] Patent: WO2013/40215, 2013, A1. Location in patent: Paragraph 00268
[2] Nucleosides and Nucleotides, 1994, vol. 13, # 10, p. 2345 - 2366
[3] Patent: WO2012/148808, 2012, A1. Location in patent: Page/Page column 78
[4] Patent: WO2018/75858, 2018, A1. Location in patent: Paragraph 0299; 0301